Regular soil testing is an important element in nutrient management. You can use soil tests as a diagnostic tool or to identify trends through time. To obtain meaningful test results, you must sample soil correctly, at the same time each year, and you must maintain records.
Soil testing laboratory methods vary, which may influence results and sufficiency ranges. Therefore, the sufficiency ranges in this publication are accurate only for the test methods listed.
Soil tests are used to measure soil nutrients that are expected to become plant-available. They do not measure the total amounts of nutrients in soil. Measurements of total nutrient content are not useful indicators of sufficiency for plant growth, because only a small portion of the nutrients are plant-available. Roots take up plant-available nutrients as positively or negatively charged ions from the soil (table 1).
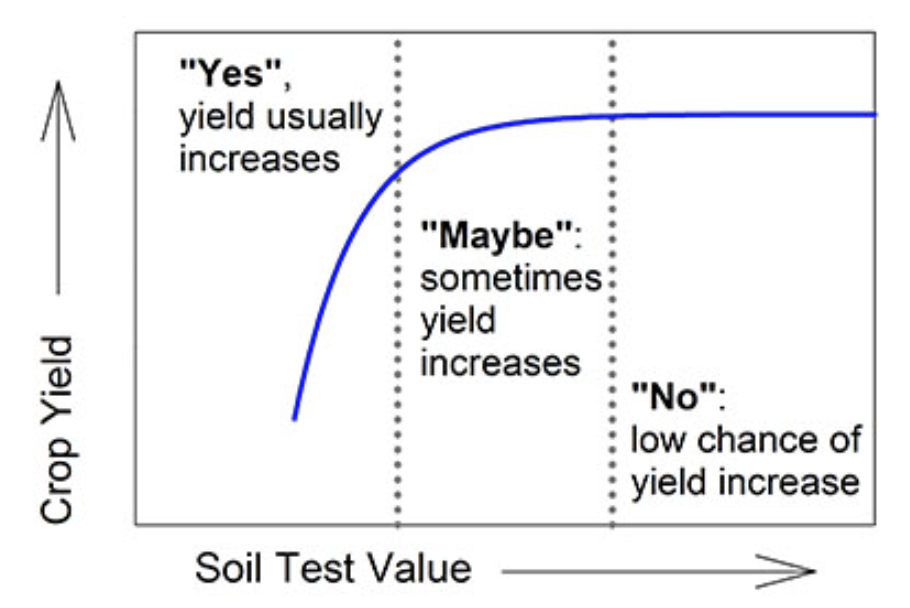
Soil test results (see figure 1) can be viewed in three categories: (1) low or yes, a fertilizer addition will likely increase growth and yield; (2) high or no, a fertilizer addition will not likely increase growth or yield; and (3) intermediate or maybe, a fertilizer addition may increase growth or yield. Categorization of soil test results into “yes,” “no,” and “maybe” assists understanding the limits and benefits of using soil test results for making nutrient recommendations.
Differing fertilizer recommendation philosophies can provide multiple recommendations from the same soil test results. For example, nutrient application recommendations of land-grant university Extension services in the western United States are based on the philosophy that nutrients should only be applied when an economic yield increase is likely. In contrast, another philosophy for nutrient application is soil test level maintenance, commonly used in the midwestern United States. Soil test level maintenance is accomplished by fertilizing nutrients removed by crop harvest. Nutrients are applied even though the soil test level for these nutrients may be sufficient.
| Nutrient | Form used by plant |
|---|---|
| Cations (+) | |
| Nitrogen | NH4+ |
| Potassium | K+ |
| Calcium | Ca2+ |
| Magnesium | Mg2+ |
| Manganese | Mn2+ |
| Copper | Cu2+ |
| Zinc | Zn2+ |
| Anions (-) | |
| Nitrogen | NO3- |
| Phosphorus | H2PO4- and HPO42- |
| Sulfur | SO42- |
| Boron | H3BO3 and H2BO3- |
| Molybdenum | HMoO4- and MoO42- |
| Chloride | Cl- |
Nutrient concentrations vary with soil depth, which affects soil test results. To determine the proper sampling depth, you must consider the purpose of the soil test. To estimate fertilizer requirement prior to planting, sample soil to the depth where most root activity will occur, usually a depth of 6 to 12 inches. Shallow sampling, 2 to 3 inches, is sometimes used to evaluate fertilizer need in perennial crops where fertilizers have repeatedly been applied to the soil surface. A combination of sampling depths may be necessary to diagnose problems in orchards.
For more information, see Evaluating Soil Nutrients and pH by Depth in Situation of Limited or No Tillage in Western Oregon (EM 9014).
Most soil test values do not vary greatly from year to year. However, some soil and environmental conditions cause fluctuations in measurements such as pH and nitrate-nitrogen. Drastic changes in test values year to year may indicate an unrepresentative soil sample or a laboratory error. When in doubt, submit a new sample or ask the lab to repeat the analysis.
Quality control programs for agricultural soil testing laboratories include North American Proficiency Testing. This program is overseen by the Soil Science Society of America and is designed to promote quality, reproducible soil testing methods.
This publication provides general guidelines for interpreting soil test results. Guidelines for specific crops are available in many other publications. If a nutrient management/fertilizer guide for the crop in which you are interested is available, use it instead of the general information in this guide. Nutrient management/fertilizer guides for Christmas trees, wheat, sweet corn, silage or field corn, peppermint, blueberries, caneberries, several grasses grown for seed, carrots grown for seed, cranberries, hops, pastures, tree fruits and nuts, alfalfa, and onions are available from OSU and PNW Extension Publishing through the OSU Extension Catalog. For potato nutrient management recommendations in Oregon, use Washington State University’s Nutrient Management Guide: Central Washington Irrigated Potatoes (EB1882).
Nitrogen (N)
Plant-available nitrogen (nitrate and ammonium)
The plant-available forms of nitrogen are ammonium-N (NH4 -N) and nitrate-N (NO3 -N)The abbreviation NH4 -N means nitrogen in the ammonium form, and NO3-N means nitrogen in the nitrate form. Soil concentrations of (NO3 -N) and NH4 -N depend on biological activity and therefore fluctuate with changes in conditions such as temperature and moisture. Nitrate is easily leached from the soil with high rainfall or excessive irrigation. Soil tests can determine NO3 -N and NH4 -N concentrations at the time of sampling but do not reflect future conditions.
The nitrogen mineralization test estimates seasonal N supply from a sample collected for wheat during late January. This test is only used for soft white winter wheat production in western Oregon.
Failure to account for NO3 -N in the soil or from irrigation water can lead to over-application of nitrogen fertilizers. Proper irrigation increases N use efficiency and reduces nitrate leaching.
Keep samples for N analysis cool and deliver them to the laboratory the day they are collected or send them using next-day delivery. If sample results are not needed immediately, samples can be frozen and shipped later.
Ammonium-nitrogen (NH4-N)
Ammonium-nitrogen usually does not accumulate in the soil, as soil temperature and moisture conditions suitable for plant growth also are ideal for conversion of NH4-N to NO3-N. Ammonium-nitrogen concentrations of 2–10 ppm are typical. Soil NH4-N levels above 10 ppm may occur in cold or extremely wet soils, when the soil contains fertilizer from a recent application when soil pH is very high or low, and when soluble salts (measured by electrical conductivity) are high.
Nitrate-nitrogen (NO3-N)—West of the Cascade Mountain Range
Nitrate-N is mobile in H2O and is a difficult test to interpret in western Oregon. Nitrate moves with the water when rainfall exceeds evaporation, usually November through April. Consequently, nitrate remaining in the soil after harvest can leach during winter rains, contaminating surface and ground-water. Situations for which nitrate-N can be used for management decisions are limited. They must be selected carefully to match crop growth and likelihood of sufficient nitrate to reduce fertilizer need. For example, midseason measurement of soil nitrate is used for silage and sweet corn production.
For more information, see Silage Corn Production Nutrient Management Guide (Western Oregon) (EM 8978); Sweet Corn Nutrient Management Guide (Western Oregon) (EM 9010).
Post-harvest soil nitrate testing can be used in some cropping systems to determine whether N supplied from all sources (fertilizer, irrigation water, organic amendments) was excessive. Interpretation of post-harvest soil nitrate analyses is crop specific. If post-harvest nitrate levels are consistently high, reduce fertilizer N inputs in future growing seasons.
For more information, see Post-harvest Soil Nitrate Testing for Manured Cropping Systems West of the Cascade Mountain Range (EM 8832).
Nitrate-nitrogen (NO3-N)—East of the Cascade Mountain Range
In arid regions, soil nitrate is evaluated by measuring NO3-N, usually in one-foot depth increments, to a depth of 2 to 5 feet. The nitrogen measured with this test is used to determine the N fertilizer application rate. Consult the nutrient management guide for an individual crop to determine sampling depth and method for crediting soil nitrate-N. If test results are reported in ppm, convert to lb/acre using table 13.
Total nitrogen
Total nitrogen analysis measures N in all organic and inorganic forms. Total nitrogen does not indicate plant-available N and is not the sum of NH4-N + NO3-N. Total N is not used for fertilizer recommendations.
A typical agricultural soil in the Willamette Valley contains about 0.10 to 0.15 percent N, or approximately 5,000 lb N/acre in the surface foot. Only 1 to 4 percent of this total N becomes plant-available (converts via microbial activity from organic form to inorganic form) during a growing season. East of the Cascade Mountain Range, soils usually have half or less this amount of total N.
Phosphorus (P)
Phosphorus fertilizer recommendations are based on a documented relationship between crop yield and extractable soil test phosphorus. Historical soil test calibration data in Oregon have been developed in relation to two specific phosphorus extractions methods: the Bray P1 test and the Olsen or sodium bicarbonate test (table 2). The Bray P1 extraction method is used for soils west of the Cascade Mountain Range, and the Olsen sodium bicarbonate (NaHCO3) extraction method is used for soils east of the Cascade Mountain Range. Soil testing laboratories also use several other extraction methods. For interpretation of results from other extraction methods, contact the laboratory that performed the analysis. Soil test P extraction methods other than Bray P1 and Olsen often have little or no field calibration studies performed to document their utility in Oregon.
| West of Cascades Bray P1 test P (ppm) | East of Cascades Olsen test P (ppm) | Recommendation (lb P2O5/acre) | |
|---|---|---|---|
| Low | <20 | <10 | 0-300 |
| Medium | 20-40 | 10-25 | 0-200 |
| High | 40-100 | 25-50 | 0-30 |
| Excessive | >100 | >50 | 0 |
Phosphorus soil tests are an index of P availability (low, medium, high, excess). When interpreting a P soil test, you must be aware of previous P management practices. Phosphorus is relatively immobile in soil. If phosphorus has been applied in a fertilizer band, concentrations of P may persist where the band was placed, making interpretation of soil test data difficult.
The phosphorus application rate necessary to correct P deficiencies varies depending on soil properties and crop grown. Phosphorus availability decreases in cool, wet soils. In many situations, banded phosphorus applications are more effective than broadcast applications, especially when soil test P values are low.
Phosphorus applications generally are not recommended when test results are high or excessive. High soil phosphorus combined with P movement from soil into surface waters can cause excessive growth of vegetation, damaging aquatic ecosystems.
Cations: potassium, calcium, and magnesium
Of the three primary cations (potassium, calcium, and magnesium), potassium requires the most management attention. Few crops have increased growth or yield from calcium and magnesium additions in the Pacific Northwest.
The soil test ranges in tables 3 and 4 are for the ammonium acetate or the sodium bicarbonate (NaHCO3) extraction method.
Potassium (K)
Excessive soil potassium levels can result in elevated K levels in grass forage crops, which may be detrimental to animal health. Conversely, very low soil test K levels can reduce plant growth. K applications in table 3 are applied as a broadcast application.
| Extractable or soil test K | Recommendation (lb K2O/acre) | |
|---|---|---|
| Low | <150 ppm* (<0.4 meq/100 g soil) | 100-300 |
| Medium | 150-250 ppm (0.4-0.6 meq/100 g soil) | 60-250 |
| High | 250-800 ppm (0.6-2.0 meq/ 100 g soil) | 0 |
| Excessive | >800 ppm (>2.0 meq/100 g soil) | 0** |
* For ammonium acetate or sodium bicarbonate extraction method.
** When extractable K is excessive, determine soil and irrigation water electrical conductivity.
Calcium (Ca)
Calcium deficiencies usually are found only on very acidic soils. They can be corrected by liming with calcium carbonate (CaCO3). Calcium is rarely deficient when soil pH is adequate. Calcium deficiency can occur at otherwise adequate soil pH values in serpentine soil (high Mg). These soils are found in the Siskiyou Mountains and other parts of southwest Oregon.
Magnesium (Mg)
Magnesium deficiencies on acidic soils can be corrected by liming with dolomitic lime (calcium-magnesium carbonate [CaCO3-MgCO3]). If the soil pH is sufficient, Mg can be supplied with Epsom salts (MgSO4) or potassium-magnesium sulfate (K2SO4·2MgSO4). Excess magnesium can occur on serpentine soils in southwest Oregon. Agricultural production on serpentine soil requires management not addressed in this publication.
| Extractable or soil test Mg | Recommendation (lb Mg/acre) | |
|---|---|---|
| Low | <60 ppm (<0.5 meq/100 g soil) | 10-100 |
| Medium | 60-300 ppm (0.5-2.5 meq/100 g soil) | 0-60 |
| High | >300 ppm (>2.5 meq/100 g soil) | 0 |
Sulfate-sulfur (SO4–-S)
Plants absorb sulfur in the sulfate form. In high-rainfall areas west of the Cascade Mountain Range, sulfate is readily leached, and soil tests are not well correlated with plant growth. In arid regions east of the Cascades, soil test information may be useful. Irrigation water may contain significant amounts of sulfate-sulfur. Plant analysis, especially a nitrogen-sulfur (N:S) ratio, is useful for diagnosing a sulfur deficiency.
| Soil test sulfate-S (ppm) | Recommendation (lb S/acre) | |
|---|---|---|
| Very low | <2 | 30-60 |
| Low | 2-5 | 10-30 |
| Medium | 5-20 | 0-20 |
| High | >20* | 0 |
*When sulfate-S soil test values are high, determine soil and irrigation water electrical conductivity.
Micronutrients
Deficiencies of micronutrients other than boron and zinc are uncommon. The availability of most micronutrients decreases as pH increases (except for molybdenum, which becomes more available as pH increases). Micronutrient deficiencies rarely occur when the soil pH is below 6.5.
Soil testing for micronutrients other than boron and zinc is recommended only when a deficiency is suspected. If you suspect a micronutrient deficiency, plant tissue testing may be a better diagnostic tool than soil testing. Soil testing will help determine what rate to apply.
Boron (B)
Crops such as alfalfa, table beets, brassica crops, caneberries, and root crops have responded to boron fertilization on B-deficient soils in western Oregon. Tree fruits and alfalfa are examples of crops sensitive to low boron levels east of the Cascade Mountain Range.
While low levels of boron may limit plant growth, high concentrations can be toxic. When applying boron, apply it uniformly as a broadcast application and mix it thoroughly with the soil.
| Soil test B (ppm) | Recommendation (lb B/acre)* | |
|---|---|---|
| Very low | <0.2 | 1-3 |
| Low | 0.2-0.5 | 0-3 |
| Medium | 0.5-1 | 0-1 |
| High | 1-2 | 0 |
| Excessive | >2** | 0 |
* Do not apply B in a concentrated area such as a fertilizer band.
** When soil test B is excessive, determine soil and irrigation water electrical conductivity and B in irrigation water.
Zinc (Zn)
A zinc soil test above 1.5 ppm using the DTPA extraction method is sufficient for most crops. Corn, beans, grapes, hops, onions, and deciduous fruit trees are especially sensitive to low levels of soil test Zn. Fertilizer applications with zinc sulfate are typically 5 to 15 lb Zn per acre.
Copper (Cu)
Copper values above 0.6 ppm using the DTPA extraction method are sufficient. Copper deficiencies are extremely rare, regardless of soil test results. The only deficiencies that have been identified are on muck soils such as those in the Klamath and Salem (Lake Labish) areas in Oregon and the Colville area in Washington. Routine copper application to mineral soil can cause copper toxicity.
Manganese (Mn)
Adequate soil test Mn varies with crop. Soil test values between 1 and 5 ppm using the DTPA extraction method are usually sufficient. Manganese deficiencies generally occur only when soil pH is 8.0 or above.
Details of Mn deficiency, Mn application, and soil pH adjustment can be found in Managing Manganese Deficiency in Nursery Production of Red Maple (EM 8905).
Manganese toxicity is more common in acidic soil than Mn deficiency. Garlic and onions are the most sensitive crops grown in Oregon. Manganese toxicity causes incomplete filling of garlic bulbs when the soil pH is below 6.5. In contrast, wheat growth and yield is not limited by Mn until soil pH is below 5.2.
Diagnosis of either Mn deficiency or toxicity should use soil pH and tissue Mn concentration in combination with soil test Mn.
For alkaline soils east of the Cascade Mountain Range, acidified microzones, such as the area around fertilizer bands or granules, can increase Mn availability. These acidified microzones can alleviate the Mn deficiency sometimes encountered on soil with high pH.
Iron (Fe)
Soil testing for iron is not recommended. Most test methods do not discern between forms of iron and therefore have little meaning for plant nutrition.
Iron deficiencies are uncommon on acidic soils in the Pacific Northwest. Where deficiencies occur, they often are associated with plants adapted to acidic soil, such as blueberries, azaleas, or rhododendrons, growing on soil with an unsuitably high pH. Acidifying the soil with elemental sulfur usually will correct Fe deficiency for these plants.
See Acidifying Soil for Blueberries and Ornamental Plants in the Yard and Garden West of the Cascade Mountain Range in Oregon and Washington (EC 1560); Acidifying Soil in Landscapes and Gardens East of the Cascades (EC 1585). For large scale agricultural production, see Acidifying Soil for Crop Production West of the Cascade Mountains (EM 8857); Acidifying Soil for Crop Production: Inland Pacific Northwest (PNW 599).
Iron salts (such as iron sulfate) applied to alkaline soil do not remain plant-available for long enough to be an effective plant nutrient source. Chelated iron fertilizers are more effective in alkaline soils than inorganic fertilizer materials, but they are very expensive. The cost usually prohibits use. Lowering soil pH to increase iron availability on a field scale is not economical. However, adding acidifying materials such as elemental sulfur to fertilizer mixtures can acidify microzones around the fertilizer material and increase Fe availability.
Turf often will become darker green when a complete fertilizer containing Fe is applied. The change in color is in spite of adequate soil test iron values.
Molybdenum (Mo)
Soil Mo concentrations are too low for most labs to evaluate. Molybdenum deficiencies are rare. Molybdenum-deficient legumes produced on acidic soil appear chlorotic or nitrogen deficient. In this situation, applying lime usually increases soil pH and alleviates the chlorosis.
Livestock producers desiring information about molybdenum in forage should have the Mo content of forage determined.
Chloride (Cl–)
Soil testing for chloride is not a common practice, and little data exist for interpretation of test results. Wheat sometimes benefits from a chloride application. Little information exists on chloride soil test values and recommended rates in Washington and Oregon. Chloride is supplied with irrigation water and from organic sources such as manure and compost. Chloride fertilizer application may occur incidentally when Cl salts are used to supply potassium or magnesium. For example, a 100 lb application of muriate of potash (potassium chloride [0-0-60]) contains 60 lb K2O and 45 lb Cl.
| Soil test Cl (ppm) | Recommendation (lb potassium chloride [0-0-60]/acre) | |
|---|---|---|
| Very low | 0-5 | 0-150 |
| Low | 5-10 | 0-150 |
| Medium | 10-20 | 0-50 |
| High | 20-50 | 0 |
| Excessive | >50* | 0 |
* When soul test CL is excessive, determine soil and irrigation water electrical conductivity and B in irrigation water.
pH, lime requirement (LR)
Soil pH expresses soil acidity. Most crops grow best when the soil pH is between 6.0 and 8.2.
Soil pH can be increased by liming, the application of calcium carbonate (limestone). The soil pH test indicates if lime is needed. The lime requirement test determines how much lime is needed.
Lime rates in eastern Oregon are much lower than in western Oregon, typically 500 to 1,500 lb/acre compared to 2,000 to 6,000 lb/acre. The lower rates reflect sandy soil or a low cation exchange capacity (CEC). The low lime rates in eastern Oregon are applied with greater frequency than lime in western Oregon, making the long-term rates for the same crop comparable.
Table 8. Soil pH ranges:
- Strongly acidic: <5.1
- Moderately acidic: 5.2–6.0
- Slightly acidic: 6.1–6.5
- Neutral: 6.6–7.3
- Moderately alkaline: 7.4–8.4
- Strongly alkaline: >8.5
SMP lime requirement test
Refer to individual crop nutrient management or fertilizer guides for lime application rate. The information provided here is very general. SMP stands for Shoemaker, MacLean, and Pratt—the people who developed the test.
Accurate lime recommendations cannot be made solely on the basis of soil pH. The SMP test is used to estimate the lime required to raise the pH of 6 inches of soil. The SMP test is performed by mixing soil with a solution buffered at pH 7.5 and determining the resulting pH. The soil’s exchangeable hydrogen or “reserve acidity” lowers the pH of the SMP solution. Soils with low SMP values have high reserve acidity and high lime requirements. Conversely, soils that are acidic and have a high SMP buffer have a low lime requirement.
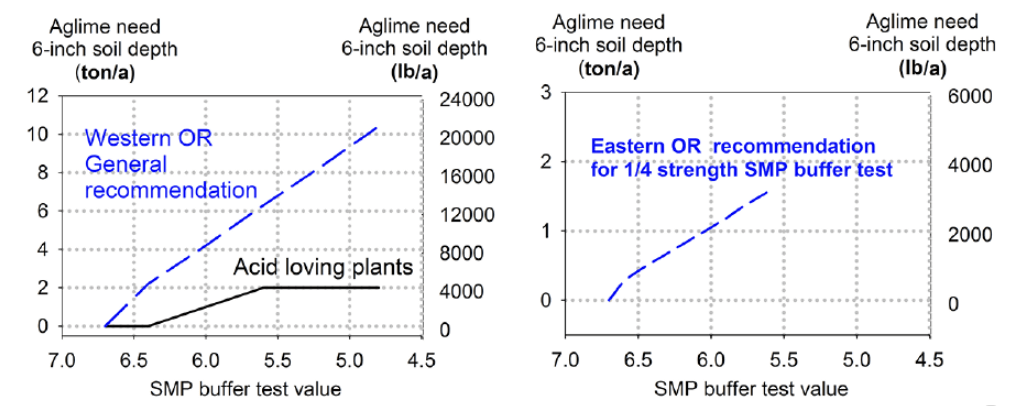
Figure 4 shows the relationship between SMP test and lime recommendation. Lime rate increases as the SMP test value decreases.
Table 9 is used to determine the amount of lime required, based on the SMP test, to raise soil pH to a desired level. The target pH is determined by the crop to be grown and possibly by other factors such as disease management. For example, when the SMP test value is 6 (left column), the recommended rate of lime for most crops in western Oregon is 8,400 lb/acre. For the same SMP test value, only 1,000 lb aglime/acre is recommended for acid loving crops like blueberries. The recommendations in table 9 assume that lime is incorporated by tillage to a depth of 6 inches.
Some soils may have a low pH (below 5.3) and a fairly high SMP test value (above 6.2). This condition can be caused by the application of fertilizer. In this case, the low pH value is temporary, and the pH of the soil will increase as the fertilizer completes its reaction with the soil.
In western Oregon, sandy soils also may have a low pH and high SMP buffer value. This condition occurs because sandy soils have low amounts of reserve acidity due to low CEC. In such cases, a light application of lime (2,000 to 4,000 lb/acre) should suffice to neutralize soil acidity. This amount of lime should be applied regardless of the SMP test value.
| SMP test result | Acid-loving plants (blueberry rhododendron, etc.) | Western Oregon field crops, horticultural production and pasture | Eastern Oregon field crops, horticultural production and pasture |
|---|---|---|---|
| 6.7 | 0 | 0 | 0 |
| 6.4 | 0 | 4,400 | 1,100 |
| 6.0 | 1,000 | 8,400 | 2,100 |
| 5.6 | 2,000 | 12,600 | 3,150 |
| 5.2 | 2,000 | 16,600 | |
| 4.8 | 2,000 | 20,800 |
*For specific recommendations, refer to individual crop nutrient management or fertilizer guides.
| When the SMP test result is... | Apply this amount of lime (lb/1,000 ft2) |
|---|---|
| <5.5 | 250 |
| 5.5-6.0 | 150-250 |
| 6.0-6.5 | 100-150 |
| >6.5 | 0 |
Sodium (Na)
Sodium is not a plant nutrient and therefore is not necessary for plant growth. High levels of sodium are detrimental to soil structure, soil permeability, and plant growth, a problem encountered east of the Cascade Mountain Range.
Sodium levels are evaluated based on exchangeable sodium percentage (ESP). The ESP is the percent of the CEC occupied by Na.
ESP values above 10 percent are of concern. Excessive sodium levels can occur naturally or can result from irrigation with high-sodium water. Reclamation involves establishment of drainage followed by gypsum application and leaching with low-sodium water.
Soluble salts
Soluble salt problems usually are associated with arid regions in eastern Oregon and Washington. Soils with high levels of soluble salts are called saline soils. Soils high in sodium are called sodic soils (see “Sodium” section). Saline-sodic soils are high in both soluble salts and sodium. Soluble salts are measured by electrical conductivity (EC) of a saturated paste soil extract.
Because salts move readily with water, salt problems can be transient. Salt toxicity can occur, and salts may leach before soil is tested. Low salt values, therefore, do not always rule out salt toxicity as a cause of problems.
Salt tolerance varies greatly among plant species. Seedlings are especially sensitive to high salt concentrations. Excessive salts may inhibit seed germination. Excessive fertilization and poor irrigation water quality are sources of salts.
See Managing Salt-affected Soils for Crop Production (PNW 601).
| EC (mmhos/cm)* | ppm salt** | Suitability for crop production | |
|---|---|---|---|
| Low | <1.0 | <640 | Suitable |
| Medium | 1.0-2.5 | 640-1,600 | Marginal |
| High | >2.5 | >1,600 | Poor, unsuitable for many crops |
*mmhos/cm is equivalent to deci-siemen/ m (dS/m) and milli-siemen/cm (mS/cm).
** Multiply mmhos/cm by 640 to estimate ppm salt.
Note: Some laboratories determine EC using a 1:1 or 2:1 ratio of water to soil, which provides a substantially lower value than the saturated paste extract. No relationship has been established for converting1:1 or 2:1 EC to saturated paste EC.
Organic matter or soil carbon
Interpretation
Soil organic matter (OM) is a surrogate for soil carbon and is measured as a reflection of overall soil health. When monitored for several years, it gives an indication whether soil quality is improving or degrading. Soil OM is important to a wide variety of soil chemical, physical, and biological properties. As soil OM increases, so does CEC, soil total N content, and other soil properties such as water-holding capacity and microbiological activity. As OM increases, so does the ability to adsorb and reduce effectiveness of many soil-applied herbicides.
Soil OM is in equilibrium with climate, soil mineralogy, and environment, providing limited capacity to accumulate organic matter. A soil with 2 percent OM will not increase to 10 percent under normal farming or gardening practices. A 2 percent increase in soil OM (e.g., from 1 to 3 percent or from 4 to 6 percent) would be considered a large change and the maximum likely for most situations in Oregon. Soil OM could increase more than 2 percent in a garden or area receiving large amounts of compost or other organic residue.
Soil OM content is not a quantitative indicator of the capacity of soil to supply plant-available nitrogen (PAN) for plant growth even though a soil with more OM contains more total N. The timing and amount of PAN released from soil organic matter is dependent on soil temperature, moisture, and many other soil management factors. Laboratories that estimate plant-available N release based on soil OM typically do not have calibration data to support their estimates.
Methods for determining soil organic matter
Soil OM values vary with analysis method. Utility of soil OM data is dependent upon analytical method. Before soil OM is determined, determine the analytical method to be used. If you monitor OM, use the same analytical laboratory and method. Differences in results from varying methods can mask changes in OM from management.
Laboratory methods used to determine OM include the following:
- Loss On Ignition (LOI) method—sample weight loss on ignition at 360ºC–400ºC
- Combustion method—combustion of sample at 900ºC–1800ºC and direct measurement of C lost in gas
- Walkley-Black method—wet chemistry reaction of soil C with an oxidizing agent (potassium dichromate)
When choosing one of these methods, consider factors outlined in table 12.
What method is preferred?
The LOI method is the least expensive. The Combustion and Walkley-Black methods are both moderate to high cost. When only an approximate value for soil organic matter is needed, the less expensive LOI test is probably adequate.
The Combustion and Walkley-Black methods both provide high accuracy and high reproducibility; the LOI method is less accurate and less reproducible. The Combustion method is more accurate and reproducible than the LOI method because it directly measures the central component of organic matter (carbon), while LOI measures weight loss (includes organic matter plus some water adhering to soil minerals). For monitoring soil OM, the Combustion method is the most suitable test.
| Method | Test cost and availability | Suitability for soil with carbonate | Accuracy and reproducibility | Comments |
|---|---|---|---|---|
| Loss on ignition | Low cost; often included in routine soil test "package" | Suitable | Low to medium; results inconsistent | LOI value can be higher or lower than "true" value determined by direct measurement |
| Combustion* | Moderate to high cost; available as additional test upon request at many labs | Suitable after sample pretreatment to remove carbonates | High | Preferred test |
| Walkley-Black* | Moderate to high cost; not offered at many labs | Suitable | High | Test generates a toxic waste (chromium) |
*Soil C determined via Combustion or Walkley-Black method is expressed as OM via the following equation OM = C x 1.7
Cation exchange capacity
CEC is a measure of a soil’s capacity to retain and release elements such as K, Ca, Mg, and Na. Soils with high clay and/or organic matter content have high CEC. Sandy, low organic matter soils have low CEC. Soil CEC is relatively constant, so no need exists for repeated analyses.
Some laboratories estimate CEC based on soil texture, organic matter content, cations, SMP, and pH. Such estimates can be inaccurate. The “sum of bases” method for calculating CEC can give inaccurate results, especially in high pH soils containing lime. CEC is used to estimate the amount of elemental sulfur required for soil acidification in western Oregon and Washington. Acidifying soil is sometimes necessary for production of blueberries or nursery stock such as rhododendrons and red maple.
See Acidifying Soil for Crop Production West of the Cascade Mountains (EM 8857); Acidifying Soil for Crop Production: Inland Pacific Northwest (PNW 599).
Base saturation
Base saturation is the percentage of the soil CEC occupied by basic cations (calcium, magnesium, potassium, sodium) at the current soil pH value. Base saturation and CEC are nearly equivalent (base saturation is 100%) when soil pH is near 7. An acidic soil has a base saturation of about 50% when the soil pH is about 5. Since base saturation is simply an indirect expression of soil pH, it is not required for making fertilizer or lime recommendations. In acidic soils, base saturation and soil pH change simultaneously. Liming increases base saturation and soil pH. Sulfur application will reduce base saturation and pH. Base saturation is not a useful concept in alkaline soils, where base saturation equals or exceeds 100%.
| To convert column 1 into column 2, divide by | Column 1 | Column 2 | To convert column 2 into column 1, multiply by |
|---|---|---|---|
| 390 | ppm K | meq K/100 g soil | 390 |
| 200 | ppm Ca | meq Ca/100 g soil | 200 |
| 121 | ppm Mg | meq Mg/100 g soil | 121 |
| 230 | ppm Na | meq Na/100 g soil | 230 |
| 1 | meq/100 g soil | cmol/kg soil | 1 |
| 3.5** | lb/acre (1 ft depth) | ppm | 3.5** |
| 43.56 | lb/acre | lb/1,000 ft2 | 43.56 |
| 43,560 | ft2 | acres | 43,560 |
| 2.471 | acres | hectares | 2.471 |
| 2.3 | P2O5 | P | 2.3 |
| 1.2 | K2O | K | 1.2 |
** This factor depends on soil bulk density. 3.5 x ppm = lb/acre-ft when soil bulk density is 1.3 grams per cm3 (81 lb/ft3). These soils typically have a medium to fine texture. Use 4 when the soil is “sandy,” more than 60% sand.
For more information
Altland, J. 2006. Managing Manganese Deficiency in Nursery Production of Red Maple. EM 8905. Corvallis, OR: Oregon State University Extension Service.
Anderson, Nicole P., John M. Hart, Neil W. Christensen, Mark E. Mellbye, and Michael D. Flowers. 2010. Using the Nitrogen Mineralization Soil Test to Predict Spring Fertilizer N Rate for Soft White Winter Wheat Grown in Western Oregon. EM 9020. Corvallis, OR: Oregon State University Extension Service.
Anderson, Nicole P., John M. Hart, Donald A. Horneck, Dan M. Sullivan, Neil W. Christensen, and Gene J. Pirelli. 2010. Evaluating Soil Nutrients and pH by Depth in Situations of Limited or No Tillage in Western Oregon. EM 9014. Corvallis, OR: Oregon State University Extension Service.
Corp, M.K., D.A. Horneck, D. Wysocki, and L. Lutcher. 2006. Monitoring Soil Nutrients in Dryland Systems Using Management Units. EM 8920. Corvallis, OR: Oregon State University Extension Service.
Fery, Melissa, Jeff Choate, and Elizabeth Murphy. 2003. A Guide to Collecting Soil Samples for Farms and Gardens. EC 628. Corvallis, OR: Oregon State University Extension Service.
Hart, J., D. Horneck, R. Stevens, N. Bell, and C. Cogger. Acidifying Soil for Blueberries and Ornamental Plants in the Yard and Garden West of the Cascade Mountain Range in Oregon and Washington. EC 1560. Corvallis, OR: Oregon State University Extension Service.
Hart, J., D. Sullivan, M. Gamroth, T. Downing, and A. Peters. 2009. Silage Corn Nutrient Management Guide (Western Oregon). EM 8978. Corvallis, OR: Oregon State University Extension Service.
Horneck, D.A., J.W. Ellsworth, B.G. Hopkins, D.M. Sullivan, and R.G. Stevens. 2007. Managing Salt-affected Soils for Crop Production. PNW 601. Corvallis, OR: Oregon State University Extension Service.
Horneck, D., J. Hart, R. Stevens, S. Petrie, and J. Altland. 2004. Acidifying Soil for Crop Production West of the Cascade Mountains (Western Oregon and Washington). EM 8857. Corvallis, OR: Oregon State University Extension Service.
Horneck, Donald, Donald Wysocki, Bryan Hopkins, John Hart, and Robert Stevens. 2007. Acidifying Soil for Crop Production: Inland Pacific Northwest. PNW 599. Corvallis, OR: Oregon State University Extension Service.
Locke, K., D. Horneck, J. Hart, and R. Stevens. 2006. Acidifying Soil in Landscapes and Gardens East of the Cascades. EC 1585. Corvallis, OR: Oregon State University Extension Service.
Staben, M.L., J.W. Ellsworth, D.M. Sullivan, D. Horneck, B.D. Brown, and R.G. Stevens. 2003. Monitoring Soil Nutrients Using a Management Unit Approach. PNW 570. Corvallis, OR: Oregon State University Extension Service.
Sullivan, D.M., and C.G. Cogger. 2003. Post-harvest Soil Nitrate Testing for Manured Cropping Systems West of the Cascade Mountain Range. EM 8832. Corvallis, OR: Oregon State University Extension Service.