Introduction
Both grass seed growers and livestock producers benefit when animals graze on grass seed fields. But many varieties of tall fescue and perennial ryegrass contain fungal toxins harmful to livestock. Learn how to detect potentially poisonous plant material and protect your flock or herd.
Most turf varieties and some forage varieties of tall fescue and perennial ryegrass are infected with a fungal endophyte. These endophytes produce bioactive compounds that enhance plant performance. Unfortunately, some of these compounds are toxic to livestock, and problems may develop when livestock consume these grasses or their byproducts above toxicity thresholds.
With the phaseout of field burning in Oregon’s Willamette Valley during the 1990s, grass seed growers baled straw for use as livestock feed for local and international markets. This secondary industry now constitutes an important agricultural commodity. Livestock producers may unknowingly create serious herd health problems by using straw or grass seed screening materials containing toxic endophytes (Figure 1) as a direct replacement for hay.
What is an endophyte?
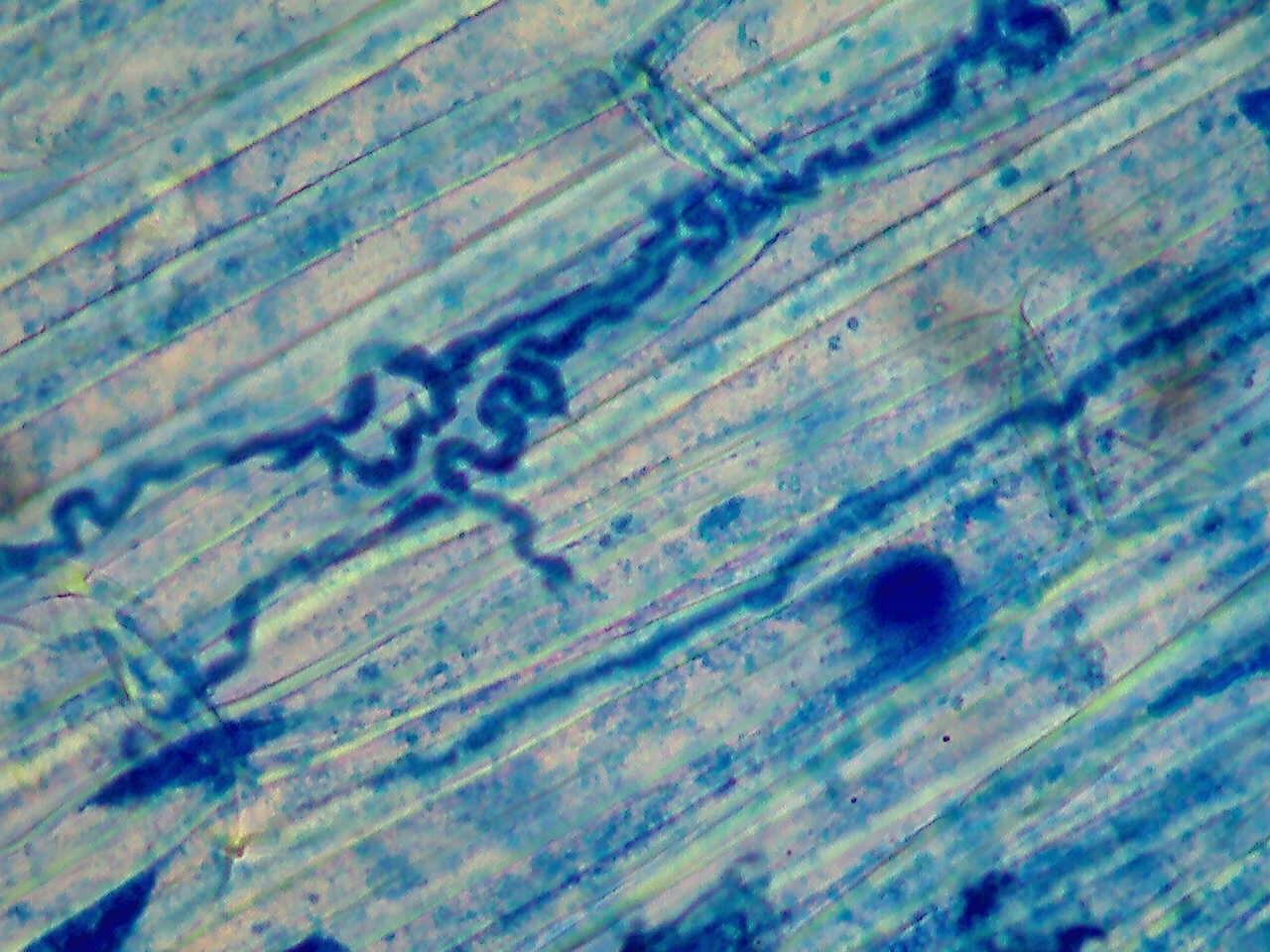
An endophyte is a fungus that lives within a plant (endo = inside + phyte = plant). The fungus grows between plant cells and into the developing seed (Figure 2). The endophyte does not affect the appearance of the plant, so only laboratory analysis can detect its presence.
Endophytic fungi are associated with both forage and turf types of tall fescue, perennial ryegrass, annual ryegrass (Lolium multiflorum), meadow fescue (Festuca pratensis), fine fescues (Festuca spp.) and orchardgrass (Dactylis glomerata).
These fungi are from the genus Epichloë. The most well-known are Epichloë coenophiala (formerly Neotyphodium coenophialum) and Epichloë festucae var. lolii (formerly Neotyphodium lolii). Tall fescue hosts E. coenophiala, while perennial ryegrass hosts E. festucae var. lolii.
Not all endophytic fungi produce levels of mycotoxins that affect livestock; however, those present in turf-type tall fescue and perennial ryegrass are usually toxic.
The relationship between a grass plant and its fungal endophyte is symbiotic; that is, they both benefit. The grass host provides nutrients for the endophyte, and the endophyte produces mycotoxins and other bioactive compounds that help protect the plant from drought stress and pests (insects and diseases). These compounds are especially beneficial in hot, dry regions.
Endophyte toxicity
The mycotoxins produced by endophytes, while beneficial to the plant, can harm livestock and birds.
Ergot alkaloids, also known as ergopeptides, are associated with fescue toxicosis. Ergovaline is the best known of the ergot alkaloids. It once was thought that ergot alkaloids were present only in tall fescue. However, a survey of Willamette Valley grass seed straw indicated that endophyte-infected perennial ryegrass also contains ergovaline. Ergovaline is now known to be produced by both E. coenophiala (in tall fescue) and E. festucae var. lolii (in perennial ryegrass).
Ergot alkaloids like ergotamine are also produced by Claviceps fungi, which can pose an additional threat to livestock (see “Ergot and Claviceps purpurea: Another source of mycotoxins,” as well as Figures 3 and 5).
Indole-diterpenes, such as lolitrem B, are associated with ryegrass staggers. These compounds are produced in perennial ryegrass by E. festucae var. lolii.
Other compounds produced by Epichloë are the lolines and peramine, which are toxic to insects but not livestock.
Endophytes in pastures
and grass seed fields
In some grass varieties, especially those grown for turf seed, many or most of the plants in a field are endophyte-infected. Turfgrass breeders often select for pest resistance and other positive qualities provided by endophytes, without being concerned about potential negative effects on livestock. Because turf varieties represent a large proportion of total grass seed production in Oregon, the risk to livestock is high when animals graze on these grass seed fields or consume grass seed straw or screening materials produced from them.
Conversely, forage grass breeders consider livestock well-being as well as forage persistence. Since the late 1970s, when the association between the endophyte in tall fescue and the livestock disorder fescue toxicosis was discovered, livestock managers avoided endophyte-infected varieties. More recently, however, forage varieties containing nontoxic endophytes have become available (see “Not all endophytes are bad”).
Some Oregon grass seed fields are planted with toxic endophyte-infected forage varieties, such as Kentucky-31. Although Kentucky-31 is widely planted, it is generally not recommended for livestock consumption, including in the Pacific Northwest.
The entire life cycle of the endophyte takes place inside the grass host. The fungus is transmitted only through the seed; a plant does not become infected from other plants. Therefore, unless endophyte-infected plants are already present, a grass pasture or grass seed field planted with a noninfected variety will be endophyte-free.
Studies show some fungicides reduce levels of fungal endophytes and ergot (see “Ergot and Claviceps purpurea: Another source of toxins”) in grass seed production fields. However, fungicide applications are not recommended in pastures or hay fields.
Not all endophytes are bad
For years, people have viewed endophytes negatively due to their potential toxicity to animals. However, not all endophytes are bad.
Research has identified endophyte strains that benefit the plant without producing mycotoxins harmful to livestock. These endophytes are known as novel, selected or livestock-friendly. Grazing grass or feeding products from these endophyte-infected plants does not adversely affect livestock.
In these nontoxic strains, natural mutations eliminate some or all of the genes required to produce a functional ergot alkaloid. Forage grasses containing these endophytes are commercially available worldwide.
Novel endophyte seed must be handled and stored properly to maintain viable endophyte presence. Avoid storing seeds in direct sunlight or in hot, humid conditions. Ideally, seed should be refrigerated. Seeds should contain at least 70% viable endophyte at the time of purchase. Plant seed soon after purchase; do not carry seed over to subsequent years, as there is a risk of losing the beneficial endophyte.
Livestock health problems associated with endophytes
Fescue toxicosis and clinical symptoms
Fescue toxicosis is associated with ergot alkaloids produced by both E. coenophiala (in tall fescue) and E. festucae var. lolii (in perennial ryegrass). These toxins are vasoconstrictors; they constrict blood vessels and reduce circulation to the body's extremities. Symptoms of fescue toxicosis include low average daily gains, reduced fertility, rough hair coats and a preference for shade or muddy areas, as animals are less able to regulate body temperature (see Figure 6 and sidebar about common clinical signs associated with ergot alkaloid toxicity).
Eventually, animals may develop fescue foot (in cold weather) or summer slump (in hot weather). However, small production losses can occur well before clinical signs appear. For example, there may be slight reductions in average daily gain, with losses becoming more severe as the level of toxin in the diet increases (see sidebar about common clinical signs associated with ergot alkaloid toxicity).
Fescue foot. This disorder is characterized by dry, dead (gangrenous) tissue in the extremities, lameness and swelling in the legs. After two or more weeks, animals develop sloughing of the hooves or lose the tips of their tail or ears. In the winter, frostbite can occur. A 10- to 20-day period of feeding on toxic endophyte-infected tall fescue or its byproducts can result in clinical signs. In the Pacific Northwest, fescue foot has occurred sporadically in cattle and sheep during the winter.
Summer slump. Hyperthermia (elevated body temperature) is the key problem associated with summer slump. Animals spend less time grazing and more time standing in water or shade in an attempt to stay cool. Other clinical signs include reduced feed intake, poor weight gain, lower pregnancy rates and decreased milk production. Poor livestock performance becomes more pronounced when temperatures exceed 70°F/21°C. In cattle, the reduction in weight gain and hyperthermia may last up to six weeks after removal from an endophyte-infected field.
In the southeastern United States, where summers are hot, summer slump is common. With more than 35 million acres of mostly toxic endophyte-infected tall fescue pasture, fescue toxicosis is the No. 1 large-animal toxicity problem in the United States. Livestock losses are estimated by the U.S. Department of Agriculture at nearly $2 billion per year for the beef cattle industry alone. The majority of this is due to decreased calving rates (failure to conceive or early embryonic loss), as well as suppressed weight gain.
Reproductive problems. Livestock that ingest ergot alkaloids can develop serious reproductive abnormalities, including failure to come into heat, early-term abortions, prolonged pregnancies, difficult births, retained placentas, poor udder development, little or no milk production, and poor newborn survival. These reproductive problems can occur in any geographic area if the feed contains toxic levels of ergot alkaloids.
Pregnant mares are especially sensitive to ergot alkaloids. Remove pregnant mares from toxic endophyte-infected pasture at least one month before foaling to prevent any negative effects. Milk production may be decreased if the mare consumes feed containing ergot alkaloids in the early stages of lactation.
Ryegrass staggers
Ryegrass staggers is caused by consumption of indole-diterpenes such as lolitrem B, which are produced in perennial ryegrass by E. festucae var. lolii. These mycotoxins are tremorgens; they cause muscle weakness, tremors and spasms. Most affected animals show no clinical signs unless they are excited. When they run, they may experience trembling, severe incoordination or falls. In Oregon, this condition is most common in sheep grazing toxic endophyte-infected perennial ryegrass as their only feed.
A seven- to 14-day exposure may induce staggers in livestock that consume contaminated material. The clinical signs usually disappear two or three days after removing animals from contaminated feed, but they can persist for up to two weeks.
|
|
||||
|---|---|---|---|---|
|
Livestock species |
No or limited clinical signs |
May see some clinical signs |
Clinical disease |
Lolitrem B (ppb)2 |
|
Horses3 |
0–150 | 150–300 | Above 300 | Not determined |
|
Cattle |
0–150 | 150–400 | Above 400 | Above 1,800 |
|
Sheep |
0–150 | 150–500 | Above 500 | Above 1,800 |
|
Camels |
Not determined | Not determined | Not determined | Above 500 |
1Ergovaline is an ergot alkaloid associated with fescue toxicosis. Some laboratories test for total ergot alkaloids, rather than for ergovaline alone.
2Lolitrem B is an indole-diterpene associated with ryegrass staggers.
3For mares in the final 60 to 90 days of pregnancy, the threshold is 0 ppb ergovaline.
Is there a safe level?
It is always better for an animal not to consume a toxin. Although livestock may tolerate some level of ergot alkaloids or indole-diterpenes, it is important to limit exposure and the concentration of toxins in feed.
Experiments and case studies conducted at Oregon State University and elsewhere have suggested threshold levels of ergovaline and lolitrem B in animal diets (Table 1). Even if animals do not show clinical disease when consuming mycotoxin levels below these thresholds, individual responses may vary. Production losses, such as reduced average daily gains, could still occur.
The thresholds in Table 1 refer to the level of toxin in the total diet, not in a single feed component. Threshold levels vary because environment and stress, as well as individual animal metabolism and length of exposure, also play roles in a particular animal developing clinical disease.
As seen above, perennial ryegrass has the potential to contain both ergot alkaloids and indole-diterpenes like lolitrem B. Animals fed endophyte-infected perennial ryegrass almost always exhibit symptoms of ryegrass staggers before symptoms of fescue toxicosis. However, some decrease in average daily gain and milk production may occur before clinical symptoms associated with ryegrass staggers appear.
Studies conducted at Oregon State University and elsewhere found that the ratio of lolitrem B to ergovaline in perennial ryegrass straw can vary from 4:1 to 10:1. Thus, perennial ryegrass with 2,000 ppb lolitrem B could contain anywhere from 200 to 500 ppb ergovaline. At these levels, ergovaline should not be a problem in sheep. But it may cause the beginning of clinical signs in some beef cattle and should be monitored in pregnant mares.
Variation in toxin concentrations
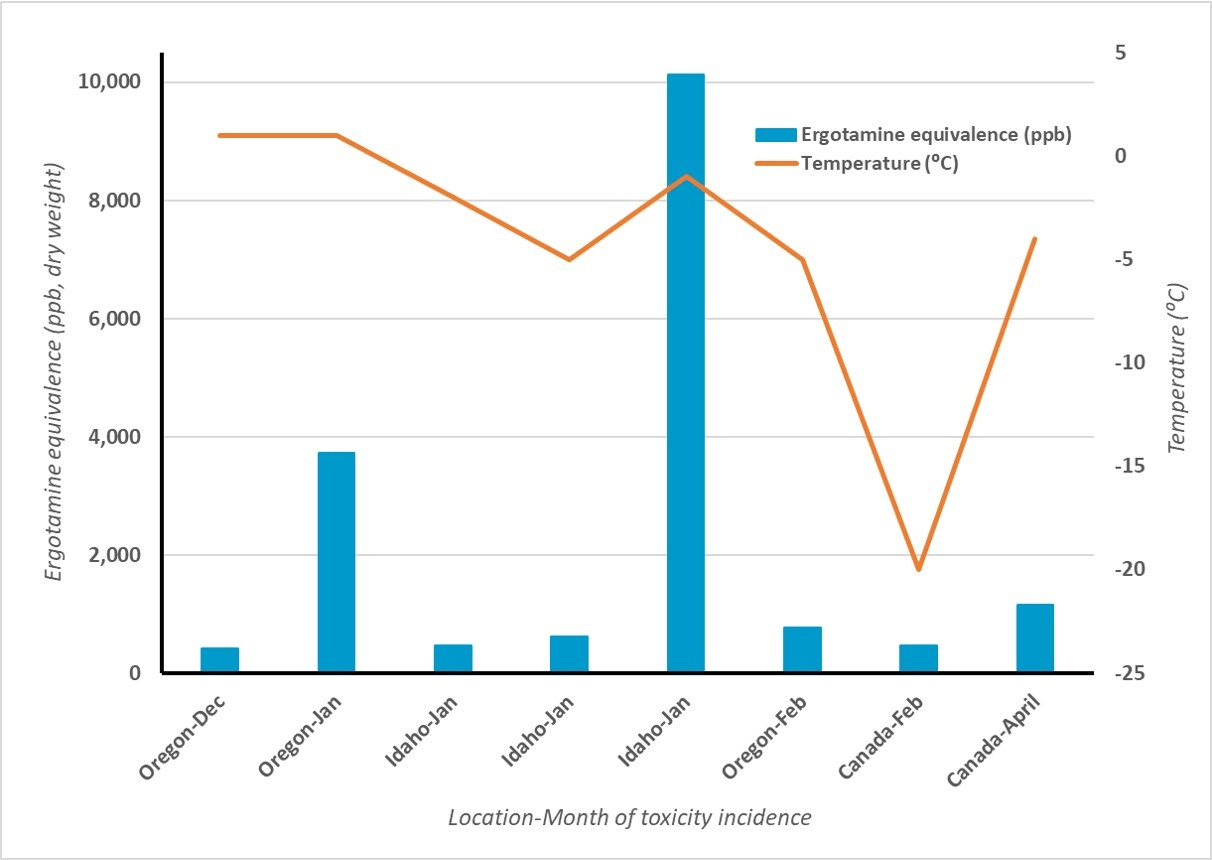
Mycotoxin levels vary across years, seasons and fields, even within the same grass variety. Variation occurs due to differences in disease and fertility management in seed production fields, seed storage conditions and environmental conditions during plant growth. The highest levels of toxin concentrations are generally found when seed heads are present in grass fields, as is typical in Oregon summers.
More ergovaline is produced when plants grow under stress. Research at Oregon State University showed that the ergovaline content in straw from the same plants was 55% lower in 1992 than in 1991. The reason was stress from above-normal spring rainfall in 1991.
Nitrogen fertilization can also influence ergovaline levels. More ergovaline is produced when soil nitrogen levels are high.
Thus, feed and forage samples should always be submitted during each harvest year for endophyte mycotoxin testing to ensure livestock safety. See “Endophyte mycotoxin testing.”
Managing forage and feed for safety
Pastures
Seed selection. Pastures planted with endophyte-free, low-endophyte or novel endophyte varieties of tall fescue or perennial ryegrass are safe to graze or cut for hay. In the Pacific Northwest, endophyte-free or novel endophyte varieties are recommended. These varieties perform well across the region’s temperate to high desert climates.
Purchase certified seed to ensure seed purity and quality. The Oregon Department of Agriculture administers the Plant Health Program, which verifies that seed is free of endophyte fungi. Tagged seed must meet the Oregon forage grass standard of 5% or less of endophyte fungi or their metabolites.
Forage and feed dilution. Forages and feed with mycotoxin levels above the thresholds in Table 1 may be fed safely, as long as they are diluted with other feedstuffs that do not contain endophyte mycotoxins. Dilution can be accomplished by interseeding endophyte-free or novel endophyte forage varieties and species into endophyte-infected grass pastures. Suitable grasses include orchardgrass, annual ryegrass and livestock-friendly varieties of tall fescue and perennial ryegrass. Legumes such as red clover, white clover and subclover are another option.
Grazing management. During the vegetative phase of grass plant growth, endophyte fungal hyphae are concentrated in the growing points (the meristematic tissue). The fungus is found first in the leaf sheaths at the base of the plant. For example, the crowns of tall fescue contain about twice as much ergovaline as the top growth. Thus, grazing grasses too short increases the risk of toxicity. Grazing crowns in a grass seed field following seed harvest is riskier than grazing the tops of plants in winter or early spring.
Grass seed straw and screening materials
As grass enters its reproductive phase and the stem elongates, endophyte fungal hyphae begin to proliferate in the developing seed heads. As a result, seed heads contain the highest concentrations of mycotoxins (Table 2).
Mycotoxins persist in grass seed straw and seed screening materials. Toxicity is not reduced by pelleting seed screenings or by making silage. Ammoniating straw can reduce the mycotoxin level, but feed should still be tested for endophyte mycotoxins after these processes to ensure it will not cause animal toxicity.
|
|
|||
|---|---|---|---|
|
Seed |
Crowns |
Tops and stems |
|
|
September–October |
2,000 | 900 | 460 |
|
December–January |
------- | 500 | 75 (tops) 200 (stems) |
1Field was 77% infected with toxic endophyte.
2Sheep grazing mostly the tops of grasses showed no signs of fescue toxicosis.
Producers feed grass seed straw and screening materials to Oregon livestock primarily in the winter. In some areas, extreme cold increases the severity of endophyte-related problems.
Feed selection and testing. Avoid feeding screening materials from unknown grass seed fields. If such feed must be used, testing for mycotoxins is strongly recommended.
Feed sources from fields known to be infected with toxic endophyte should also be tested. Since mycotoxin levels can vary greatly, testing is the best way to decide whether or how a feedstuff can be used in a livestock diet.
Stored feed. In both tall fescue and perennial ryegrass, the endophyte fungus begins to lose viability when seed is stored for 18 months or more, especially under warm, humid conditions. Novel endophytes may lose viability more quickly. Thus, the infection rate is reduced in old seed and in plants grown from old seed. However, endophyte-produced mycotoxins persist even after several years of storage, so feeding stored grass seed and screening materials is still risky.
Supplementation. Grass seed straw and screening materials are considered low-quality feed ingredients. A low-quality diet can result in poor body condition, increasing endophyte-related problems. Always supplement low-quality feed to meet nutritional needs.
Endophyte mycotoxin testing
Sampling procedures
It is important to submit a representative sample for testing. The test result received will only accurately reflect the mycotoxin concentration in a randomly sampled lot of straw or other source of feed material. You need at least 50 grams for adequate testing.
Air-dry samples out of direct sunlight. Make sure samples are completely dry before submitting; moist samples are not acceptable. See “How to take a sample” for detailed instructions.
- Hay and straw. Use a core forage sampler that has an exterior diameter of one-half inch and a sample length of at least 12 inches.
- For bales, center the core sampler at the end of the bale and drill horizontally. For each lot, take one core sample each from at least 20 random bales.
- For stacks of straw, core every fourth or fifth stack or take at least five random samples from each side of the stack.
- Fields. You can sample grass plants at any time of the year. However, sampling during a vegetative stage is preferable. Choose at least 20 random locations. Clip grass plants at ground level.
- Seed screening materials or pellets. Take 15 to 20 small subsamples randomly from the storage containers. Mix thoroughly to combine into one representative sample. Send at least 3 cups (about 750 mL) of the mixed material to the laboratory.
Submitting samples
Collect samples into polyethylene freezer bags, insert the sample submission form from the Endophyte Service Laboratory website (either through the noncommercial or commercial client link), then seal and ship to Oregon State University, Endophyte Service Laboratory, Oak Creek Building, Room 139, 3015 SW Western Blvd., Corvallis, OR 97333. We can also receive samples in the laboratory if you prefer an in-person drop-off.
For more information
- Oregon State University Endophyte Service Laboratory
- Oregon State University Seed Laboratory
- Oregon Department of Agriculture Endophyte Testing and Seed Tagging Program
- Oregon Tall Fescue Commission
- Oregon Ryegrass Commission
- Oregon State University Tall Fescue Online Monograph
- Oregon Seed Growers League
References
Alabdouli, K.O., L.L. Blythe, J.M. Duringer, A. Elkhouly, A. Kassab, M. ASkar, E.E. Mohammed, A. Al-Juboori and A. M. Craig. 2014. Physiological effects of endophyte-infected perennial ryegrass straw on female camels in the Middle East. Emirates Journal of Food and Agriculture 26(1): 82-92.
Bourke, C.A., E. Hunt and R. Watson. 2009. Fescue-associated oedema of horses grazing on endophyte-inoculated tall fescue grass (Festuca arundinacea) pastures. Australian Veterinary Journal 87(12):492–498.
Craig, A.M., L.L. Blythe and J.M. Duringer. 2014. The role of the Oregon State University Endophyte Service Laboratory in diagnosing clinical cases of endophyte toxicoses. Journal of Agricultural and Food Chemistry 62:7376–7381.
Craig, A.M., J.L. Klotz and J.M. Duringer. 2015. Cases of ergotism in livestock and associated alkaloid concentrations in feed. Frontiers in Chemistry 3(8):1–6.
Duringer, J.M., L.L. Blythe, C.T. Estill, A. Moon, L. Murty, S. Livesay, A. Galen and A.M. Craig. 2021. Determination of a sub-chronic threshold for lolitrem B and perennial ryegrass toxicosis in Angus cattle consuming endophyte-infected perennial ryegrass (Lolium perenne) straw over 64 days. Livestock Science 250: 104570.
Hovermale, J.T., and A.M. Craig. 2001. Correlation of ergovaline and lolitrem B levels in endophyte-infected perennial ryegrass (Lolium perenne). Journal of Veterinary Diagnostic Investigation 13:323–327.
Johnson, L.J., A.C. de Bonth, L.R. Briggs, J.R. Caradus, S.C. Finch, D.J. Fleetwood, L.R. Fletcher, D.E. Hume, R.D. Johnson, A.J. Popay and B.A. Tapper. 2013. The exploitation of Epichloae endophytes for agricultural benefit. Fungal Diversity 60(1):171–188.
Kaur, N., S.C. Alderman, D.L. Walenta, K.E. Frost, J.K.S. Dung and P.B. Hamm. 2016. Evaluation of new fungicide chemistries and application strategies to reduce ergot in grass seed production systems. In 2015 Seed Production Research Report. N. Anderson, et al. (eds.). CrS 152. Oregon State University.
Murty, L.D., J.M. Duringer, A.M. Craig. 2018. Co-exposure of the mycotoxins lolitrem B and ergovaline in steers fed perennial ryegrass (Lolium perenne) straw: Metabolic characterization of excreta. Journal of Agricultural and Food Chemistry 66:6394-6401.
Parish, J.A., J.R. Parish, T.F. Best, H.T. Boland and C.A. Young. 2013. Effects of selected endophyte and tall fescue cultivar combinations on steer grazing performance, indicators of fescue toxicosis, feedlot performance, and carcass traits. Journal of Animal Science 91(1):342–355.
Takach, J.E. and C.A. Young. 2014. Alkaloid genotype diversity of tall fescue endophytes. Crop Science 54(2):667–678.
Young, C.A., N.D. Charlton, J.E. Takach, G.A. Swoboda, M.A. Trammell, D.V. Huhman and A.A. Hopkins. 2015. Characterization of Epichloë coenophiala within the U.S.: Are all tall fescue endophytes created equal? Frontiers in Chemistry 3(8):40.
Young, C.A., D.E. Hume, and R.L. McCulley. 2013. Forages and Pastures Symposium. Fungal endophytes of tall fescue and perennial ryegrass: Pasture friend or foe? Journal of Animal Science 1:91(5):2379–2394.