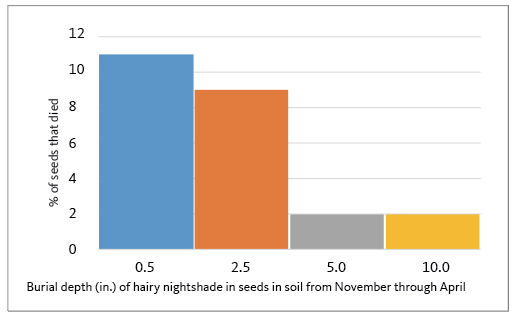Snap beans are often one of the more profitable crops in a row crop rotation. Weeds in snap beans reduce yield and contaminate products sent to processors or markets. Even a few weeds surviving until harvest can make mechanical harvest difficult. However, yield losses due to weed competition or load rejection at the processing plant can be minimized.
Whether organic or conventional, a proactive approach that encompasses the entire crop rotation is justified. This approach should:
- Strive to prevent weed seed introductions or seed production, and reduce seed survival throughout the crop rotation (particularly for weeds such as nightshade);
- Use a diversity of methods to control weeds in the crop to avoid crop loss and development of herbicide-resistant weeds; and
- Make sure that herbicides or other practices used to control weeds in snap beans do not carry over to other crops and jeopardize future crop production.
Prevention
Minimize introductions
Successful weed management prevents new weeds from entering a field and gaining a foothold. Weed seedling identification is important so that new introductions can be quickly spotted and the source identified. Prevention strategies should target and minimize seed production of surviving weeds.
A particularly important time to destroy weeds is after crop harvest. Weeds are commonly observed growing in fields after crops are harvested in late summer and fall, allowing weeds to continue to produce viable seeds. The fall season is particularly important for weeds such as hairy nightshade. This plant continues to produce berries (and seeds) as long as stems are allowed to grow, and the longer the season, the more seeds are produced.
A good case study that highlights the need for prevention is the increase of puncturevine in the Willamette Valley over the last decade (Figure 1). Seeds are transported from the east side of the Cascade Mountains on equipment (mostly tires) and end up in staging areas in vegetable production fields or along roadsides. Checking tires on harvest equipment before unloading it into fields is essential. If puncturevine is sighted, it is best to dig up the plant and destroy it by burning. Puncturevine seed dormancy is long. Researchers in eastern Washington demonstrated that maximum emergence after seed was produced occurred 3 years after the initial seeding event. (For more information, see “References,” Boydston, 1990).
Another example relates to the introduction and expansion of velvetleaf. Velvetleaf was first spotted in Yamhill County in 1928 and probably arrived in cottonseed meal from the south, which was fed to cattle. In 2002, velvetleaf seed was found in manure from poultry farms near Aurora, Oregon. Until recently, these introductions seldom threatened crop production, but velvetleaf populations have increased to the point where they are threatening production of row crops in some areas of the Willamette Valley (Figure 2). It is important to determine the source of the seed and, if already established in fields, consider removing seedlings before they can produce seeds. Imazamox (Raptor) and bentazon (Basagran) are commonly applied postemergence to control weeds in snap beans, but they have little, if any, effect on velvetleaf.
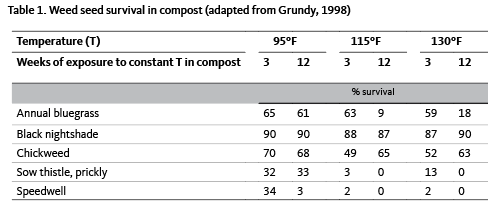
Weed seeds are often introduced in biomass or compost brought onto farms. If using animal manures or compost to fertilize crops, learn where or how the forage was produced, and whether weed seeds may be present. If using animal manures, remember that many weed seeds pass undamaged through the digestive system of ruminants. If using compost, make sure that compost temperatures exceed 160ºF for at least 48 hours so that all weed seeds are killed.
In reality, 160ºF is difficult to uniformly obtain during the composting process. Many weed seeds will die if exposed to 140ºF for at least 60 minutes. Some weed seeds will succumb to temperatures as low as 95ºF if the duration is long enough (Table 1). Others, such as black nightshade, will be unaffected by 130ºF even when exposed for 12 weeks. Common purslane has seeds that are difficult to kill at 160ºF, even after extended periods of time.
Managing weed seed survival
Leaving weed seeds on the soil surface in the fall reduces their winter survival. This exposes weed seeds to seed predators (such as mice, ground beetles, and birds), more extreme temperature variation, and wetting and drying. Weed seeds usually lose viability more quickly if on or near the soil surface compared to when they are buried. In contrast, soil tillage in the fall that buries weed seeds may enhance survival. In a 4-year study in the Willamette Valley, tillage during direct-drilling of a cover crop in the fall enhanced wild proso millet weed emergence the following year, mainly because the no-till drill caused enough soil disturbance to bury and protect seeds during the winter. In another study, nightshade seed survival was less when the seed remained near the soil surface rather than when buried 5 or 10 inches during the winter (Figure 3).
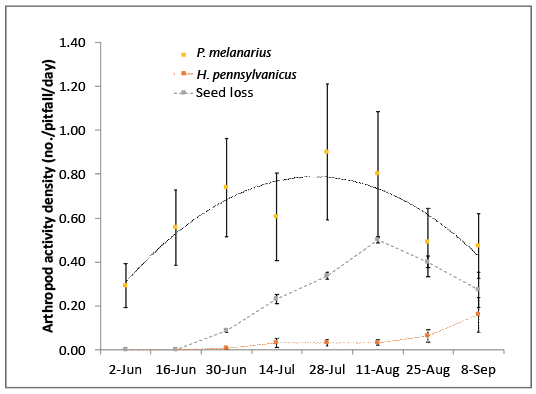
Many organisms eat weed seeds. In contrast to mice and voles, carabid beetles generally do not threaten crop seeds unless the seeds are exposed on the soil surface. Carabid beetles are common in most cropping systems in the Willamette Valley and, based on pitfall trapping over 3 years at more than 25 sites, Pterostichus melanarius far outnumbered other carabid beetles (Figure 4). These beetles consume weed seeds that range in size from very small pigweed seeds to large seeds like wild proso millet. However, these beetles are completely inept at locating weed seeds that are covered by soil.
Even though these beetles typically consume a variety of foods (omnivorous), they often prefer to eat small larvae, earthworms, and slug eggs rather than weed seeds. There are exceptions. P. melanarius will seize wild proso millet seeds and bury them immediately if they are the only food choice available (Figure 5). It is unclear why they bury the seeds or what happens to the seeds after burial.
In several on-farm experiments in the Willamette Valley, it was noted that broad-spectrum insecticides such as chlorpyrifos increased the consumption of weed seeds by carabid beetles, probably because the insecticide removed preferred insects. The question remains whether cropping systems should be designed to improve habitat and encourage larger populations of P. melanarius. Even though weed seed loss from seed stations mirrored carabid beetle activity and density (Figure 6), the number of seeds taken was very low, and there was no determination whether the seeds were eaten or buried. (For more information, see “References,” Brainard, et al., 2013; Green 2010; and Moulton 2011).
In some cases, spring primary tillage (tillage before crop planting in the spring) can be used to manage the number of weeds that emerge in the spring. This is especially the case if, in the previous year, weeds were allowed to produce seeds in a field that traditionally had few weeds. Weed seeds can be plowed under with inversion (moldboard) plows so they cannot emerge in the spring. Few weed seeds produce seedlings if buried more than 2 inches deep; the zone of optimum emergence for most small-seeded summer annuals is ¾ inch or less, depending on soil moisture and texture, and seed size. Tillage following spring plowing must be relegated to the near-surface soil layer to make this strategy work.
This strategy can backfire if proper records are not maintained and weed seeds are mistakenly brought to the surface with deep tillage in the spring. Seeds that are moved to 6 inches or below in the soil will typically survive longer than seeds that are buried near the soil surface. Deep plowing to bury crop residues may bring these same seeds to the surface 7 to 10 years later.
Crop rotation
Crop rotation is the cornerstone of a successful weed management program. A well-constructed rotation will prevent a shift to one or a few dominant weed species and mitigate the development of herbicide resistance. Crop rotations that alternate between winter and summer crops, competitive and uncompetitive crops, high-fertility crops and low-fertility crops, or irrigated and nonirrigated crops will select for different weed species. The more variation that is built into a crop rotation, the less likely a single weed species will begin to dominate. For instance, winter annual crops, such as winter wheat, host an entirely different suite of weeds than irrigated summer annuals. Even though summer annual weed seeds remain ungerminated in the soil during the wheat crop, they lose viability over time.
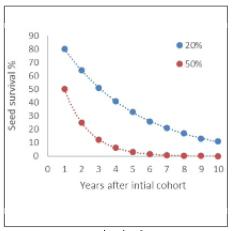
As Figure 7 demonstrates, a 3-year break in a rotation will substantially reduce summer annual broadleaf seed survival, particularly if weed seed mortality is 50 percent per year. Many summer annual weeds, including pigweed, common lambsquarters, and the nightshades, have mortality rates within the 50 percent range in fields that are tilled annually. Even with 20 percent loss of seed viability per year, only 11 percent of the seed remains after 10 years.
Of course, the other advantage of a well-developed crop rotation is that it often improves snap bean growth and reduces the incidence of diseases like root rot and white mold.
Cover crops
Another proactive approach is the use of winter or summer cover crops. Keeping the soil covered during the winter will help prevent the establishment of winter weeds such as shepherdspurse, deadnettle, speedwell, annual bluegrass, annual ryegrass, and other perennial grasses that might interfere with future crops in the rotation. Competitive spring and summer cover crops will even keep patches of perennial weeds (such as nutsedge) from expanding in area. It is important to seed at rates high enough to establish a solid stand of cover crop. For cereals, 45 seeds/ft2 is usually adequate or about 1.5 times more than when seeding for grain.
Other important cultural practices
Fertilize the beans, not the weeds, when possible. Most summer annual weeds are extremely well adapted to high nitrogen soils and will do much better, comparatively, than beans under high fertility conditions. Banding fertilizers when possible will limit the competitive ability of weeds that are far from the nitrogen band and reduce the number of weed seeds produced.
Wider row spacings result in less in-row area to manage weeds, higher plant densities in the row that are more competitive with weeds in the row, and less white mold (for more information on the effect of row spacing on white mold in snap beans see “References,” Peachey, et al., 2006).
A competitive crop is essential. Planting beans when soil temperatures are optimum ensures quick emergence and good competition with emerging weeds. Snap bean canopies also close faster when beans are planted in fields with adequate rotation intervals, especially if 5 or more years occur between bean crops. A 5-year rotation interval significantly reduces root diseases such as Fusarium and will allow better plant growth and yield.
Weed control methods
Once a proactive and preventive weed management plan is in place, focus can then turn to managing weeds in the snap bean crop. While herbicides are essential in conventional bean production, herbicides seldom provide all of the control required by producers, especially if weed seeds are abundant in the soil. Other strategies will be needed to supplement herbicides to meet weed control objectives.
Weed-free periods
An important concept to keep in mind when managing weeds is the weed-free period. The critical weed-free period is the minimum length of time a crop should be kept weed-free from the time of planting to avoid a yield reduction from weed competition. Weed-free periods have been established for many crops and range from 3 weeks in short-season crops to 8 weeks in crops like carrots. Weed-free periods are often measured in growing degree-days to improve precision for early and late plantings. Other factors that influence the critical weed-free period are the density and species of weed seeds present in a field at planting. The critical weed-free period for snap beans is typically 2 to 4 weeks.
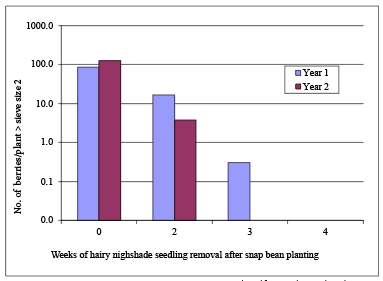
The weed-free concept can be extended to weeds such as hairy nightshade that not only compete with the crop for water, light, and nutrients but also produce berries that may contaminate the harvested product. Two years of research in western Oregon indicated that removal of hairy nightshade after planting was needed for up to 3 weeks in 1 year and 4 weeks the following year to prevent plants from producing berries that were greater than sieve size 2. (For more information, see “References,” Boydston and Peachey, 2004). Nearly the same result was obtained over four planting dates from May 4 through June 20 (Figure 8).
Stale and false seedbeds
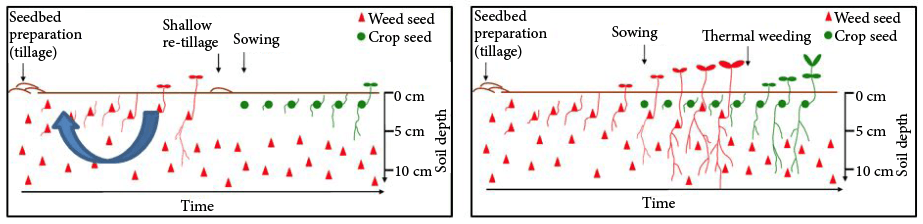
Stale and false seedbeds (Figure 9) can be used to reduce weed density in the crop and are particularly useful in organic production. Success is dependent on conditions that cause weed seeds to germinate: moist soil and temperatures sufficient to cause germination of the desired species. The tillage event used to stimulate weed emergence must be planned to coincide with normal times of weed emergence. Otherwise, tillage before weeds are ready to germinate brings a fresh set of weed seeds to the soil surface, where they may germinate after the false or stale seedbed is completed.
One challenge of using stale seedbeds is timing them to coincide with the emergence of targeted weeds. Another challenge is that stale seedbeds give symphylans (a root-feeding insect, Scutigerella immaculata) a foothold. While symphylans are not a huge problem in snap beans, increasing the interval between deep tillage and planting the crop gives symphylans an opportunity to cause serious damage to many smaller-seeded crops. One of the most effective controls for symphylans is to till soil deep and vigorously and then immediately plant the crop. (For more information, see “References,” Umble, 2006).
False seedbeds are developed by tilling the soil well ahead of snap bean planting, followed by shallow tillage or other techniques to destroy emerged weeds before the crop is planted. This technique may be applied several times in succession to exhaust germinable weed seeds at the soil surface (Figure 9).
Stale seedbeds are developed by tilling the soil before crop planting to stimulate weed emergence, but the tools used to remove the weeds are applied after the crop is planted and before the crop emerges.
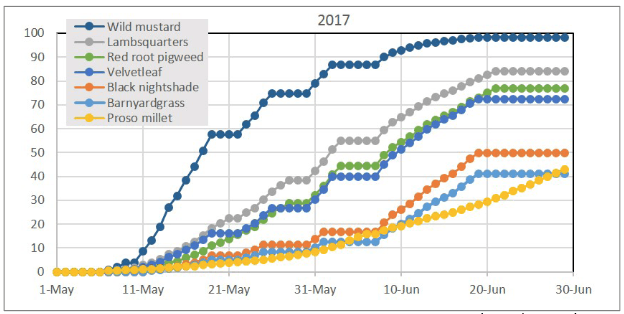
The success of stale and false seedbeds is dependent on careful timing. They work best when weeds are most likely to germinate, which does not occur at the same time for all species. Therefore, it is essential to know which weeds are present and when they will have the highest germination potential. For example, data from weed emergence models such as Weed Cast, using Willamette Valley weather data, predicted the emergence of seven common weed species following seedbed preparation on May 1, 2017, in Corvallis, Oregon would fall into three groups (Figure 10). Wild mustard emerges first; common lambsquarters, redroot pigweed, and velvetleaf are in in the second cohort; and black nightshade, barnyard grass, and wild proso millet emerge last. This sequence of emergence is relatively consistent over years, as long as ample moisture is available. If moisture is sufficient, soil temperature becomes the main, driving factor.
It is clear from this model and field-based observations that using a stale seedbed may be effective for weeds like wild mustard because a high percentage of seedlings emerge in a short time. On the other hand, using a stale seedbed to control proso millet may be very difficult because seedling emergence is slow and will continue long into the planting season. The results shown in Figure 10 are based on mathematical estimates, but these estimates appear to confirm what has been observed in western Oregon for wild proso millet—continued emergence throughout the growing season.
Techniques used to kill weeds in stale seedbeds after they emerge include flame weeding, blind tillage (cultivating after seeding but before the crop emerges), or burndown herbicides. If blind tillage is used, the soil surface must be level and uniform. A bar is run over the seed row and just under the soil surface with gauge wheels to control depth. It may be best to use independent gangs that only target the area over the row and have a gauge wheel attached to ensure a precise depth of cultivation. Blind tillage is particularly suited to large-seeded crops like snap beans that can be planted deep. Planting depth may need to be adjusted to accommodate blind tillage.
Cultivation
Many cultivation methods can be used, but effective strategies usually move soil away from the row very early and then move it back toward the row later. Ridges left during cultivation must be compatible with harvesting equipment. Withholding irrigation immediately before and after cultivation will improve the effectiveness of cultivation. Also, less frequent irrigation tends to favor large-seeded crops over small-seeded, shallow-rooted weeds.
Care should be taken to avoid bringing new weed seeds to the surface during cultivation. Most weed seedlings emerge from seeds that are buried less than 1 inch deep. Larger seeds, such as wild proso millet and barnyardgrass, may emerge from as deep as 2 inches. As much as 30 to 40 percent of that zone may be depleted if tillage and weather conditions coincide to produce favorable weed seed germination conditions in the spring. Cultivation that moves soil from below 2 inches may bring new soil to the surface with a new set of weed seeds. This is less of a concern as the season progresses.
The first cultivation should occur when weeds are in the white thread stage (radicle has emerged) or just beginning to poke through the soil. If the crop is starting to emerge, this is a precarious time to cultivate, but it can be very effective. If the soil is slightly crusted, many seedling cotyledons may be sheared off during slight shifting of the soil. Some cultivation equipment is designed to do this, such as torsion weeders. Check the stability of snap bean plants in the row during cultivation by gently tugging on them to make sure that the radical was not disconnected from the soil. Precision guidance systems significantly improve the effectiveness of most cultivation equipment.
The equipment types available for cultivation in row crops are numerous and beyond the scope of this publication. Figure 11 shows the three types of cultivation equipment that might be considered for use.
The rotary hoe is a “preemergence” weeder that is typically used over the entire field just before or during crop emergence. Large-seeded crops or cereals are most tolerant of this type of weeding, while small-seeded weeds are kicked out of the soil by the rotating tines. Proper timing and soil moisture are critical. The height of the tines must be adjusted so that they are above planting depth or they may cause too much crop damage. Gauge wheels are preferred over simple three-point hitches without gauge wheels.
Spring tine weeders represent a second category of cultivation equipment; the differential between crop and weed size determines effectiveness. Soil textures and soil moisture also affect efficacy. Seeding rates should be raised to accommodate seedlings that are damaged or destroyed during cultivation. For more information on the wide diversity of cultivation equipment available, see the video “Vegetable Farmers and Their Weed Control Machines” at https://articles.extension.org/pages/18436/video:-vegetable-farmers-and….
Finger weeders are a “postemergent” cultivator that can only be used between rows and after crops emerge. The fingers are positioned so they reach into the crop row and loosen soil next to crop plants while blades or sweeps follow the spyders and uproot small weed seedlings next to the row. The differential between crop and weed size and how strongly the crop is rooted determines weed removal efficacy and how well the crop tolerates cultivation. The use of precision guidance systems such as RTK or inline row sensors to align the cultivator with the crop row will significantly improve cultivation accuracy and crop safety, and allow faster cultivation speeds.
In addition to the above mechanical cultivators, several automated weeders and thinners are in development in other vegetable production areas of the U.S. These cultivators use row guidance systems and computer-aided detection of weeds and crops to thin crops like lettuce or remove weeds. (See video at https://bit.ly/2M4VvC7.) These have been developed for wide in-row spacings typical of broccoli, cauliflower, corn, and squash. It is unclear how these technologies might be adapted for crops like snap beans that have much greater in-row densities. One possibility would be in organic systems where nonselective herbicides could be targeted in the near-row area to avoid crop damage and reduce the cost of the herbicide and hoeing.
Herbicides for conventional and organic production
Herbicides are commonly used in conventional systems because the potential return on investment is very high. Uncontrolled weeds can be very competitive and can substantially reduce yield. Cultivation and hand hoeing are very expensive and time-consuming. Processors also have a very low tolerance for contamination, and in the case of hairy nightshade berries, the tolerance is zero. The threat of having a load of snap beans rejected at the processing plants encourages growers to use every tool possible to reduce the risk of contamination.
Using preemergence herbicides
Three principles should guide the use of preemergence, soil-applied herbicides.
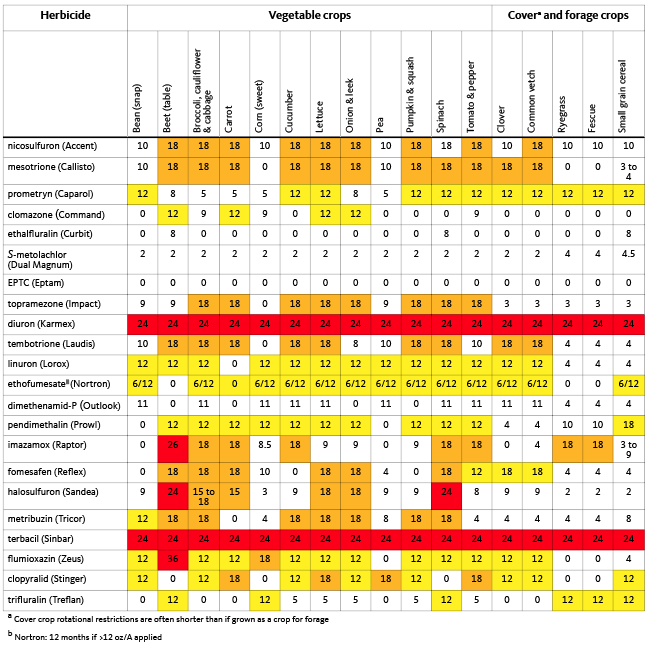
1. Apply the herbicide before weed seeds germinate. Many herbicides are not effective if applied after weed seeds germinate. The radicle of the seed emerges first, then quickly grows down and away from the herbicide-treated zone. If the herbicide is not active when the radicle emerges, the weed seedling may survive. Herbicides such as S-metolachlor, Group 13 (Dual Magnum); dimethenamid-P (Outlook), and trifluralin, Group 3 (Treflan) provide very poor control if the weed seeds have germinated. This is not the case for all soil-applied herbicides, including oxyfluorfen (Goal) and flumioxazin (Chateau), but even in these cases, the smaller the seedling, the better the efficacy. To ensure proper preemergence herbicide efficacy, till the soil, then apply and incorporate the herbicide before weed seeds have a chance to germinate. (See Table 2 for predicted efficacy.)
2. Activate the herbicide. Activation of soil-applied herbicides distributes the herbicide so that it comes in contact with the weed seed. Water usually works best to activate preemergence herbicides, but tillage is required in some situations because of herbicide volatility (such as with EPTC, Group 8, commercially known as Eptam) or because the herbicide is very insoluble and is difficult to incorporate with irrigation or rainfall (Treflan). Water carries the herbicide into the soil and distributes the herbicide throughout the soil. The herbicide must be in contact with the seed when it germinates.
3. Preserve the chemical barrier. Properly applied and activated herbicides present a barrier to weed seeds. Too much irrigation water or rain on a soluble herbicide such as Dual Magnum or Eptam will destroy the chemical barrier; the herbicide is washed too far down and becomes diluted at the soil surface. Incorporating the herbicide too deeply into the soil is another way to diminish the effective chemical barrier. Mechanical disturbance also can destroy this barrier and allow weed seedlings to escape. Fomesafen, Group 14 (Reflex) and lactofen, Group 14 (Cobra) are preemergence herbicides that kill some weeds on contact when weed seedlings grow into the chemical barrier at the soil surface. If the herbicide concentration is lessened at the soil surface because the surface layer has been moved (by cultivation, tillage, or foot traffic), the herbicide barrier will be broken or diminished, and weed control will suffer.
If water is being used to incorporate an herbicide, it is typically done before the crop emerges. Irrigation as the crop emerges through S-metolachlor or lactofen will often cause substantial crop injury, but usually will not compromise yield (Figure 12).
Common herbicides available
Preplant or post-plant
Eptam has been an industry standard over the decades. Eptam is preplant incorporated (PPI) to control many grasses and some broadleaves, including nightshade. (See Table 2 for efficacy rating.)Frequent use of Eptam will lessen its effectiveness because of increased microbial activity in the soil. It should be incorporated shortly (minutes) after it is applied to the soil. This is particularly important if the soil surface is moist.
Treflan is PPI primarily for grass control, but is also for lambsquarters and pigweed. Treflan does not control nightshade weeds, however. Activity may linger into the fall and cause damage to seeded grasses.
Dual Magnum is applied PPI and post-plant surface (PPS) to control annual grass and broadleaved weeds. Nightshade and lambsquarters control is typically less than 70 percent, especially if too much rain falls or irrigation water is applied after planting.
Halosulfuron, Group 2 (Sandea) is a sulfonylurea herbicide and ALS inhibitor used at rates of ¾ oz/A or less. Sandea has no effect on nightshade, but it is a good tank mix with Eptam to improve control of lambsquarters and nutsedge. This herbicide has long rotation intervals for crops in the beet family and for many Brassica and mustard crops.
Cobra is a protoporphyrinogen oxidase (PPO) inhibitor that provides exceptional control of hairy and black nightshade. This herbicide must be incorporated with rain or irrigation water within 48 hours of planting to avoid crop damage. Intense rainfall as the beans emerge will sometimes cause soil treated with Cobra to splash onto plants (Figure 13). Application rates should be adjusted for areas of fields that are sandy or gravelly. Cobra is very active on soils with low cation exchange capacities (CEC) and may damage emerging snap beans.
Reflex is a selective preemergence herbicide for broadleaf weed control in snap beans. The active ingredient, fomesafen, is a PPO inhibitor in the same group of herbicides (mode of action Group 14) as Cobra, Goal, SureGuard, Chateau, and Spartan. Reflex has both soil and foliar activity, but it is only labeled for preplant surface (PREPS) and post-plant surface (PPS) use in snap beans. Reflex provides exceptional control of pigweed, nightshade, and common purslane; acceptable control of common lambsquarters; and suppression of nutsedge at the 1 pt/A rate.
Reflex should be applied at 1 pt/A PREPS or PPS. If applying PPS, apply Reflex well before snap bean seeds have germinated or seedlings have begun to emerge. If applying PREPS, weed control in the seed row may be diminished where double disks have moved the soil. A tank mix of Dual Magnum plus Reflex, both at 1 pt/A, will control most annual broadleaf and grass weeds (Figure 14).
Like many other preemergence herbicides, Reflex herbicide must be activated with about ⅓ to ½ inch of water after it is applied to maximize efficacy. Lesser amounts of rainfall (⁵/₁₀₀ inch) can still be very effective if weeds have begun to emerge when the rainfall occurs. However, in typical situations, irrigation or rainfall of ½ inch after application carries the herbicide into the soil and next to the weed seed where it controls the weed after germination. Failure to incorporate Reflex herbicide increases the risk of damage to the crop if there is a heavy downpour as the snap bean seedlings emerge. Downpours just as the crop is emerging will cause the most damage to the crop; activating the herbicide within 48 hours after planting will alleviate some of this risk. Snap beans are more tolerant of Reflex than Cobra in these situations.
Reflex herbicide is persistent, especially in dry climates. Read the label carefully. There are clear directions for the crops that can be planted after using Reflex. Research at Oregon State University indicated no injury to winter wheat planted 4 months after application, or cabbage and sweet corn planted 12 months after application. However, injury to beets was visible at the 2 pt/A rate 12 months after Reflex was applied. Bentgrass also was injured when planted in the fall 4 months after Reflex was applied to squash, even though Reflex is not typically recognized as a grass herbicide. Because of the potential for carryover and injury to other crops, Reflex use is limited to one application every other year and a maximum of 1 pt/A per season.
Postemergence herbicides
There are only two herbicides labeled for postemergent (POST) control of broadleaves: imazamox, Group 2 (Raptor) and bentazon, Group 7 (Basagran). The Raptor label requires that it be tank-mixed with Basagran to improve crop safety and improve the spectrum of control. Raptor does not control lambsquarters, and the addition of Basagran will significantly improve control.
The label rate of Basagran to tank mix with Raptor ranges from 6 to 16 oz/A. As mentioned above, Basagran improves crop safety when Raptor is applied to beans. However, tank mixing Basagran with Raptor will also reduce the efficacy of Raptor on moderately susceptible weeds such as pigweed. Therefore, depending on the suite of weed species found in a field, applicators should consider fine-tuning Basagran rates to maximize weed control. If lambsquarters dominate the field, the full 16 oz/A rate of Basagran is recommended. If pigweed is the primary species, applicators should consider reducing the Basagran rate to 6 oz/A.
Basagran can also be used to suppress and sometimes control Canada thistle and nutsedge. This requires two applications at 1 lb ai/A each, separated by 10 to 12 days.
Sandea is labeled for postemergence weed control at the second to fourth trifoliate growth stage, but it will typically cause yellowing of the snap beans, slow growth, and in some cases reduce yield. There are few situations where Sandea is beneficial, unless nutsedge is completely out of control and a reduction in yield can be tolerated. Directed applications will significantly reduce the potential of crop injury.
Sethoxydim (Poast), clethodim (Select Max), and quizalofop (Assure)—all of which are Group 2—control grasses in snap beans. Drought-stressed plants or plants that have been disturbed with cultivation equipment will not be controlled. Fescue is particularly difficult to control with these herbicides. Some grass species may be resistant to this group of herbicides in the Willamette Valley.
Managing selection pressure and herbicide resistance
Overuse of a single herbicide or herbicides that have the same mode of action in a plant can lead to two different outcomes: either a shift to a weed species that the herbicide does not control or herbicide resistance
An individual herbicide generally is not equally effective on all weed species. If the same herbicide is applied year after year, the weeds present in a field may slowly shift to a species that is not controlled by that herbicide, and at that point the herbicide becomes useless. Good examples of this are Treflan and Sandea that have no effect on hairy nightshade, or grass herbicides such as Poast and Select that have no effect on broadleaf weeds. Continued use of these herbicides will increase the abundance of these weeds if weed seedlings are not controlled and are allowed to produce seeds.
Another possibility is that the applied herbicide may begin to select for resistant plants of one species that differ slightly in genetic makeup compared to the wild or normal type. Herbicide-resistant plants occur in a population in extremely small numbers. Repeatedly using one herbicide allows these genetically unique plants to survive and reproduce. In some ways, this is like a new species that has invaded the field. If an herbicide previously controlled a particular weed and now does not, then herbicide resistance may be the culprit.
There are many examples of herbicide-resistant weeds in the Willamette Valley, but at this point these weeds do not have a large impact on weed control outcomes in snap beans. Annual ryegrass is resistant to several herbicides in some areas of the Willamette Valley, including glyphosate and Group 1 herbicides such as sethoxydim (Poast). This may have implications for managing this species in snap beans.
Herbicide resistance is known to develop very quickly for some herbicides like Poast (Group 1 herbicide) and Raptor or Accent (Group 2 herbicides). Raptor is applied to more than 50 percent of the snap bean acreage in the Willamette Valley each year. Black nightshade has been documented to have become resistant to Group 2 herbicides in other areas of the U.S. (Table 3), and producers should take steps to mitigate resistance in Oregon. (See “Preventing selection of herbicide-resistant weeds”.) Herbicide resistance is less likely to develop in herbicide families that include Dual Magnum (Group 15) and Eptam (Group 8).
Preventing selection of herbicide-resistant weeds
1. Herbicide rotations. Do not apply the same herbicides year after year. Rotate among herbicide groups with different sites of action.
2. Use short-residual herbicides (herbicides that do not persist in soil for long periods). Using short-residual herbicides that are not applied repeatedly within a growing season reduces the selection of herbicide-resistant weeds.
3. Apply imazamox (Raptor) to small weeds. There are 158 weed species worldwide that are resistant to Group 2 (ALS inhibitor) herbicides like Raptor. This herbicide is an extremely effective nightshade herbicide when it is applied to small weeds; large weeds are much more difficult to control. Every stewardship effort should be taken to ensure that this herbicide remains effective in snap beans in the future because it has a very low impact on humans and the environment.
4. Crop rotation. Each crop is produced under a unique suite of practices, including herbicide use. Crop rotation usually diversifies herbicides and other practices used to control weeds.
5. Use integrated practices and nonchemical methods to manage weeds. Cultivation, for example, can be a very effective tool for eliminating weed escapes that may represent the resistant population.
6. Monitor fields and record weed escapes after herbicides are applied. Do not let weeds go to seed or even produce pollen, as pollen often carries a dominant resistance gene. Hand pull or hoe weeds that should have been killed by the herbicide and that did not escape because of poor application technique.
7. Prevent resistant weed seeds from entering fields via equipment. Whenever possible, finish field work last at sites that are known to have herbicide-resistant weeds, and clean equipment thoroughly before moving from field to field.
Avoiding the consequences of herbicide carryover
Herbicide carryover after snap beans or from other crops to snap beans
Herbicides are made to kill weeds, and some of the most effective herbicides persist in the soil or plant residues long enough to harm following crops (see Table 4 for a comparison of herbicide rotation restrictions). Of the herbicides used in snap beans, Reflex, Raptor, and Sandea have the longest rotation restrictions and can harm sensitive crops such as beets (Figure 15) or broccoli (Figure 16), even when planted 26 months later. Clomazone (Command) is seldom used in snap bean production but may injure small grain cereals that follow in the fall. Treflan and Dual Magnum residues sometimes linger and reduce emergence of small-seeded grasses in the fall.
The key to preventing injury from herbicide carryover is to read the label carefully, keep good records of what has been applied, and then refer to the records before planting the crop. Many mishaps could be prevented by simply reviewing herbicide application records before planting. Of the herbicides that are used in crops grown in rotation with snap beans, those of most concern are clopyralid, Group 4 (Stinger); diuron, Group 5 (Karmex); and terbacil, Group 5 (Sinbar). Stinger herbicide injury (Figures 17 and 18) is unique because the herbicide will sometimes linger in plant residues, and because it is occasionally an unexpected contaminant of compost. Karmex is often used in crops such as grass seed, a very common rotation crop with snap beans. Occasionally, Sinbar that was applied to mint has injured beans planted up to 4 years later (Figure 19). Herbicides such as Sinbar have long half-lives, and repeated applications may produce herbicide residues in the soil that are much greater than anticipated when applied over consecutive years.
Rotational restrictions for cover crops often are less stringent than for forage crops that may be grown for seed or feed, or for crops where screenings will be fed to ruminants. Rotational restrictions are set so that herbicide residues in the food or feed crop do not exceed the federal tolerance (maximum residue limit) established by the EPA when the product was labeled. Rotational restrictions are also set based on whether a crop will be injured following an application months prior. When planting cover crops, the consequence of crop injury from herbicide carryover may be much less, and occasionally registrants mention on the label that cover crops can be planted earlier than the rotational restriction as long as minor injury can be tolerated. A good example is the Treflan label, which allows much shorter rotation intervals if some injury to cover crops is acceptable.
Residue tolerance, herbicide MRLs, and marketability
The EPA labels herbicides for use in snap beans under authority of the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA). The amount of herbicide residue allowed in snap beans is determined by applying labeled rates of an herbicide in many field tests, measuring what residues and metabolites remain in the product at harvest (if any), and then comparing the data to what is known of the effects on humans, test animals, and wildlife. The acceptable level of residue in a harvested crop becomes the legal limit and is known as the “tolerance,” usually expressed in parts per million or billion. The tolerance is used to gauge what rates and at what times an herbicide can be applied to the growing crop. Tolerances are listed at the Electronic Code of Federal Regulations (https://bit.ly/2KqFOnQ).
Tolerances may also determine what herbicides can be used in a rotation, and whether potential carryover to food or forage follow-crops will exceed the federal tolerance for that crop. Rotational intervals on labels are based on both potential injury to rotation crops and potential of the crop to pick up herbicide residues.
Maximum Residue Limits (MRLs) are essentially tolerances from other countries. MRLs may or may not conform to U.S. tolerances. In some cases, MRLs for herbicides that we want to apply in Oregon may not even exist in countries where we export. Although not related to snap beans, an important example is the use of HPPD herbicides in sweet corn. MRLs have not been established by many of the Pacific Rim countries for these herbicides, thus closing a market opportunity. It is important to consult with handlers if there is doubt about whether MRLs exist for any herbicide. Consult the global MRL Database at www.globalmrl.com.
Organic herbicides
Several herbicides are approved and are available for use in organic production. Most organic herbicides are only approved for use in areas where crops are not grown (such as fence rows), for burndown before the crop is planted, or (in some cases) as burndown between the rows with shielded sprayers. These herbicides are all nonselective contact herbicides—they damage any plant tissue that they contact and will not prevent weeds from emerging from the soil (like conventional, soil-active herbicides). These herbicides are either extracted from plants (such as cloves, citrus rinds, or lemon grass) or are produced by fermentation (acetic acid or vinegar). They are used at relatively high rates (2 to 7 gal/A) and are expensive.
An example of the broadcast efficacy of these herbicides for control of summer annual broadleaves is provided in Figure 20. One advantage of these herbicides is that they have very short or no reentry intervals (REI). The main disadvantage to using these herbicides is the high cost and lack of control of grasses and perennial weeds. The lower the dilution rate, the better the efficacy on the weeds it contacts. Efficacy will be best if the total spray volume is above 40 gal/A, but on most labels the recommended spray volume rate is much higher.
One possible use of organic herbicides is in stale seedbed systems where the herbicide would be banded over the row just before the snap beans emerge. In-row weed control is a challenge in snap beans, and limiting the organic herbicide to a band over the row would greatly reduce the cost.
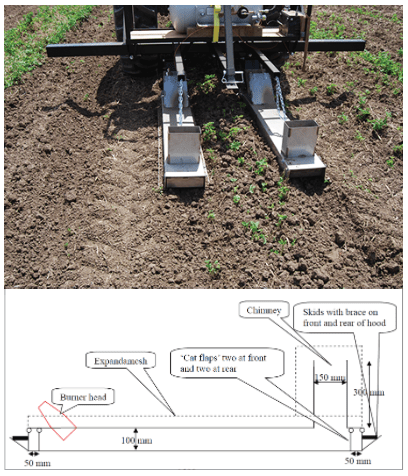
Stale seedbeds using conventional or organic herbicides and flame weeders
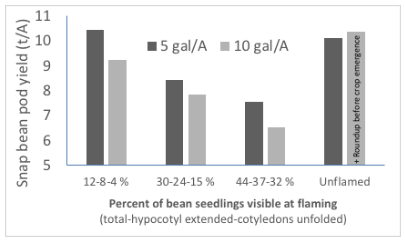
Flame weeding can be used in stale seedbed systems before the beans have emerged or when the first hypocotyl crooks become visible (Figures 21 and 22). The hypocotyl is moderately tolerant to flame intensities that will kill very small weeds. In a recent trial, flaming did not reduce plant stand, yield, or grade when applied at 5 gal/A when 12 percent of the bean seedlings were visible and 8 percent of the hypocotyls had emerged from the soil. The number of weeds present 2 weeks after flaming was 82 percent less in flamed areas than in unflamed ones. When flame was applied at a propane rate of 10 gal/A, plant stand and yield declined by 12 percent, but grade was unaffected. This data demonstrates that flame may be applied to snap beans that are emerging but that factors such as rate and environment will determine risk of crop injury. And given that the time between 0 and 10 percent emergence may be as short as 4 hours, it is important that flame applications are prompt.
Flame that is banded over the row (as shown in Figure 23) in stale seedbed systems to catch that first flush of weeds can be very cost effective ($10 to $20/A at $2/gal if applied as a band over the row), but it all depends on the timing of weed emergence. If snap beans emerge before the weed seedlings, flaming may damage the crop and do nothing to improve weed control.
The timing and amount of heat needed to kill weeds is critical to the success of flame weeding. Weeds with only cotyledons or two true leaves are much easier to kill than larger weeds, particularly if they are grasses. Inexperienced flamers often apply too much heat. Only apply enough to rupture the cells, not fry the plant. A simple test can be done by squeezing a leaf after flaming between thumb and finger. If your thumb print remains, the leaf turns a dark watery green, or is easily smashed, then sufficient heat has been applied (Figure 24). The best time to flame weeds is usually late afternoon, but this is also a time that is very difficult to see the flame. To improve safety and reliability, a repeating spark should be applied to keep the flamer lit.
Like all weed control practices, some weeds will be resistant to flame weeding, and weed shifts (changes in the abundance of species) are expected over time with continual use. Grasses are difficult to control with flame weeding because their growing point is below ground and well protected. Broadleaves are more susceptible because their growing point is above ground, but even then, if the weed is too large, it may regenerate from latent buds low on the stem that are not damaged. Common purslane is completely tolerant of flame weeding because of its fleshy leaves, even as a small seedling. If flaming is used frequently, expect weed shifts to grasses such as annual bluegrass and broadleaves such as common purslane.
Summary
Managing weeds in snap beans is no easy task, whether in conventional or organic production. The outcome of a weed management program will determine whether a crop is harvested and sent to the processing plant, and whether or not the product is rejected because of contamination. Crop rotations are the anchor to successful management, followed by steps to prevent weed seed production or new introductions to the farm. Herbicides are essential in conventional production to reduce hand-weeding costs. Herbicide efficacy may need to be supplemented with cultivation or stale and false seedbeds. Weed management plans should include strategies to ensure that hairy and black nightshade, pigweed, and other important weeds do not become resistant to imazamox, Group 2 (Raptor) herbicide.
References
Boydston, R. 1990. Time of emergence and seed production of longspine sandbur (Cenchrus longispinus) and puncturevine (Tribulus terrestris). Weed Science 38:1 pp. 16–21.
Boydston, R.A., and R.E. Peachey. 2004. Predicting hairy nightshade berry production in peas and snap beans based on thermal time of emergence and heat units. Proc. of the West. Soc. of Weed Sci. 57:55. Colorado Springs, CO.
Brainard, D., R.E. Peachey, E.R. Haramoto, J.M. Luna, and A. Rangarajan. 2013. Weed ecology and nonchemical management under strip-tillage: implications for northern U.S. vegetable cropping systems. Weed Technology 27(1):218–230.
Gonzalez-Andujar, J.L., G.R. Chantre, C. Morvillo, A.M. Blanco and F. Forcella. 2016. Predicting field weed emergence with empirical models and soft computing techniques. Weed Research. Version of record online:
10 AUG 2016.
Green, J. 2010. Structuring habitat to conserve ground beetles (Coleoptera: Carabidae) and reduce summer annual weeds in agroecosystems. MS thesis, Horticulture Dept., Oregon State University.
Grubinger, V. and M.J. Else. 1996. Vegetable farmers and their weed-control machines [DVD]. University of Vermont Extension. Available for purchase at http://www.uvm.edu/vtvegandberry/Videos/ weedvideo.htm
Grundy, A.C., M. Green, and M. Lennartsson. 1998. The effect of temperature on the viability of weed seeds in compost. Department of Crop and Weed Science, Horticulture Research, Wellesbourne, Warwick, United Kingdom.
Merfield, C. 2015. False and stale seedbeds: the most effective non-chemical weed management tools for cropping and pasture establishment. Report number 2-2015, The BHU Future Farming Centre, NZ.
Moulton, L. 2011. Using stable isotopes and visual gut examination to determine the diet composition of Pterostichus melanarius (Coleoptera: Carabidae) in Western Oregon vegetable row crops. MS Thesis, Horticulture Dept., Oregon State University.
Peachey, E. Editor. 2018. Pacific Northwest Weed Management Handbook. Corvallis OR: Oregon State University. https://pnwhandbooks.org/weed
Peachey, E., R.L. Ludy, M.L. Powelson, and D. McGrath. 2006. Modification of plant arrangement suppresses white mold of snap beans. HortScience 41:1298–1302.
Peachey, R.E. and C. Mallory-Smith. 2007. Influence of winter seed position and recovery date on hairy nightshade (Solanum sarrachoides) recruitment and seed germination, dormancy, and mortality. Weed Sci. 55:49–59
Peachey, R E. and C. Mallory-Smith. 2011. Effect of fall tillage and cover crop strategies on wild proso millet (Panicum miliaceum) emergence and interference in follow-crops. Weed Technol. 25:119–126.
Umble, J. 2006. Symphylans: soil pest management options. ATTRA Sustainable Agriculture. https://attra.ncat.org/attra-pub/viewhtml.php?id=127
(accessed April 4, 2018).
Trade-name products and services are mentioned as illustrations only. This does not mean that the Oregon State University Extension Service either endorses these products and services or intends to discriminate against products and services not mentioned.
Trade-name products and services are mentioned as illustrations only. This does not mean that the Oregon State University Agricultural Experiment Station either endorses these products and services or intends to discriminate against products and services not mentioned.
Use pesticides safely!
- Wear protective clothing and safety devices as recommended on the label. Bathe or shower after each use.
- Read the pesticide label—even if you’ve used the pesticide before. Follow closely the instructions on the label (and any other directions you have).
- Be cautious when you apply pesticides. Know your legal responsibility as a pesticide applicator. You may be liable for injury or damage resulting from pesticide use.
Extension work is a cooperative program of Oregon State University, the U.S. Department of Agriculture, and Oregon counties. Oregon State University Extension Service offers educational programs, activities, and materials without discrimination on the basis of race, color, national origin, religion, sex, gender identity (including gender expression), sexual orientation, disability, age, marital status, familial/parental status, income derived from a public assistance program, political beliefs, genetic information, veteran’s status, reprisal or retaliation for prior civil rights activity. (Not all prohibited bases apply to all programs.) Oregon State University Extension Service is an AA/EOE/Veterans/Disabled.