Commercial blueberry production in Oregon is dominated by the northern highbush type, although rabbiteye blueberries are produced on limited acreage. This publication addresses nutrient management of northern highbush blueberries in western Oregon. Where data are available to support management differences by blueberry type, this information is provided.
The following questions are addressed in this publication. These questions should be answered before any fertilizer is applied.
- How much fertilizer should be applied?
- When should the fertilizer be applied (timing)?
- What source (type) of nitrogen should be applied?
- What method of application should be used?
Blueberry plants require chemical elements from air, water, and soil to ensure adequate vegetative growth and fruit production. When levels of these nutrients in the plant are inadequate, growth and yield may be affected. Severely reduced nutrient supply can lead to visible nutrient deficiency symptoms, such as leaf discoloration (Figure 1 and 2) and distortion.
The goal of fertilization for any high-value crop is to remove limitations to yield and quality by supplying the crop with ample nutrition in advance of demand. Nutrient application should be based on soil and plant analyses and grower experience. Consider the return on your fertilizer investment as well as environmental stewardship. A fertilizer application should be "bio"logically sound; in other words, it should produce a measurable change in plant growth or nutrient status. A fertilizer application is expected to increase fruit yield or quality, producing a return on your investment.
Results from a nutrient application will vary from field to field and from year to year. This guide does not address every circumstance encountered by growers. Growers, with the assistance of county Extension faculty and field representatives, should annually consider nutrient needs of each field. Routine analysis of soil and tissue samples is helpful in determining fertilizer requirements. Annual tissue testing can detect low nutrient concentration before visible symptoms or yield reduction occurs.
To assist with interpretation of soil and tissue analyses, keep records of weather, disease problems, and nutrient applications for each field. Observations of annual growth (whip number, diameter, and shoot length), yield, leaf color, and fruit quality (berry set and firmness) are also helpful.
The use of fertilizer should be part of a complete management package. Management practices from selection of certified plants to pre- and postharvest irrigation must be performed in an appropriate and timely manner so that plants can benefit from applied nutrients. Nutrient application is not a substitute for poorly timed irrigation, late harvest, or failure to control insects, diseases, rodents, or weeds. Soil properties such as high pH and/or poor drainage can be significant limiting factors to obtaining high berry yields. Increasing fertilizer rates or adding nutrients already in adequate supply will not correct these limiting factors.
Tissue testing
By analyzing plant tissue for nutrient content (tissue testing), you can evaluate the adequacy of nutrient levels. Plant tissue analyses indicate which elements are accumulated in adequate, deficient, or excessive amounts. This information will help you decide whether fertilizer is needed and, if so, how much and what kind.
Changes in tissue analyses for some nutrients, such as nitrogen (N) and boron (B), can occur within a few weeks of nutrient application. Typically, however, changes in nutrient concentration in a perennial crop such as blueberries require a longer time (sometimes as long as 2 years), especially when immobile materials (phosphorus and potassium) are applied to the soil surface.
Tissue testing can be used for any of the following:
- Predicting fertilizer needs of annual crops
- Diagnosing problems
- Evaluating a fertilizer program for perennial crops
Tissue testing can be used to monitor and adjust fertilizer use during early growth stages of annual crops such as potatoes, sugar beets, or lettuce. A tissue test can help growers anticipate fertilizer needs for these annual crops.
In contrast, tissue test results are not very useful for predicting current-season fertilizer needs of perennial crops such as blueberries. In part, this limitation is due to the minimal short-term effect of fertilizer on yield in perennial crops. In producing blueberries, tissue testing is best used for end-of-season evaluation of a fertilizer program.
If problems such as poor growth or discoloration of shoots or whips appear during the growing season, you can use a tissue test to check for nutrient deficiencies. When sampling outside the late July to mid-August time period (see “When to sample,” below), you must collect a companion sample from an unaffected area for comparison.
Tissue testing is based on sampling at the proper time, sampling the appropriate plant part, and using “standards” for comparison. The following sections discuss these topics.
When to sample
Tissue samples should be collected when nutrient concentration is stable. Samples collected just a few days apart during periods of rapidly changing nutrient concentration can give quite different results. For example, tissue N concentration changes rapidly early in the growing season but slowly after early July (Figure 3).
For evaluation of blueberry nutrient status, collect leaf tissue between late July and mid-August. Samples collected between these dates produce consistent analytical results.
How to sample
Collect samples of the most recent fully expanded leaves (Figure 4). Do not take leaves from whips. Collect leaves that are free of disease or other damage if possible. Pick leaves so that the petiole (stem) remains with the leaf.
Sample 5 leaves each from at least 10 plants distributed randomly throughout the field. A single sample should not represent more than 10 acres.
Do not mix cultivars in a tissue sample, as mixed samples have more variability. For example, cultivars might have different fruiting seasons and times of growth flushes; thus, they might differ in leaf nutrient concentration at any given time.
If the purpose of leaf sampling is to diagnose a suspected problem, collect one sample from affected plants and another from apparently healthy plants.
Do not wash samples. Put leaves in a paper (not plastic) bag. Air dry them or send them to a laboratory immediately. To avoid spoilage, ship fresh (moist) samples early in the week to ensure delivery before the weekend.
A list of laboratories performing tissue analyses is found in Laboratories Serving Oregon: Soil, Water, Plant Tissue, and Feed Analysis (EM 8677).
Frequency of sampling
Annual sampling from all fields is recommended so that trends can be observed. This will allow adjustments in fertilizer application before a nutrient deficiency or excess occurs.
Other considerations
Tissue analysis results outside the normal range cannot always be attributed to the fertilizer program. Insufficient mineral nutrient concentration can be caused by unfavorable soil pH; saturated or dry soils; high temperatures; frost; shade; weed, insect, or disease pressure; herbicide injury; or overcropping. Insufficient pruning, resulting in a yield too high for the bush to support, will reduce overall bush vigor and root, shoot, and whip growth, and will result in tissue concentrations outside the normal range.
Several fungicides contain plant nutrients. Because tissue samples are not washed before analysis, high levels of copper (Cu), manganese (Mn), or zinc (Zn) might be the result of fungicide residue. High B and Zn also might occur after liquid or foliar fertilizer application.
Soil sampling
In contrast to use of annual tissue analyses to assess the adequacy of a fertilizer program, soil analysis is most useful before planting to determine what adjustments are needed. For example, soil pH might be too high for optimum blueberry production.
Obtain soil samples at least a year before planting so soil pH can be adjusted if necessary (see “Site preparation”). Applications of elements such as K and magnesium (Mg) also are more accessible to plant roots when mixed with the soil rather than broadcast over the surface after planting.
After planting, soil analyses can be helpful in diagnosing problems, such as high soil pH or excessive salts. Obtain samples from the area between the dripline and the crown.
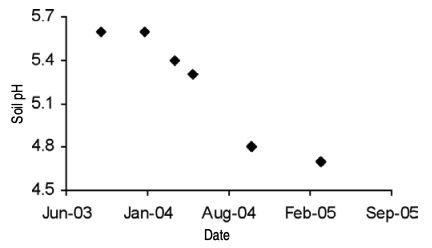
Fertilizer is commonly applied in a wide band centered on the blueberry rows. This application method concentrates nutrients, complicating soil sampling after site establishment. For example, banding of N can decrease soil pH in the row more than between rows (Figure 5). In this example, the surface soil pH also was lower than the subsurface soil pH. A similar pattern can be found for nutrients such as K when fertilizer is placed in a band on the soil surface.
Additional information is found in Soil Sampling for Home Gardens and Small Acreages (EC 628) and Monitoring Soil Nutrients Using a Management Unit Approach (PNW 570-E).
Site preparation
Planting blueberries is an investment that can cost more than $10,000 per acre (Blueberry Economics, EM 8526-E). A suitable site is necessary for growth, optimum production, and prompt recovery of the initial investment. Such a site has air and water drainage, goodquality irrigation water, and adequate soil pH. Additional information is available in Proceedings for the 2004 Blueberry Production Course and in Highbush Blueberry Production (PNW 215).
Acidifying soil
High soil pH is one of the primary problems encountered with new blueberry fields. When blueberry plants are grown in high-pH soil, the leaves, especially new growth, are yellow with green veins or completely yellow. These leaves are small and often turn brown and fall from the plant before the end of the season. Little growth occurs, and some plants may die (Figures 6 and 7).
Plants stunted by high soil pH usually do not recover even when soil pH is reduced. Blueberries established in high-pH fields often require replanting to obtain a vigorous, uniform stand.
The soil pH recommended for highbush blueberry production is 4.5 to 5.5; for rabbiteye blueberries it is 4.2 to 5.0. Use the upper limit of soil pH as your target when planting, as soil pH will decline over time with application of N fertilizer. Plant highbush types in fields with soil pH about 5.5 and rabbiteye blueberries at pH 5.0.
Measure soil pH well in advance of final site selection and planting. If soil pH is too high, acidify the soil a year before planting. Adjusting soil pH after planting is expensive, difficult, inefficient, and sometimes impossible.
Preplant acidification usually is achieved by applying and incorporating elemental sulfur (S). Elemental S can be supplied by a single application or by several smaller applications. Neither approach is superior. Splitting the elemental S application is a cautious approach compared to a more aggressive single application. Use finely ground S for acidification. Incorporation of elemental S is necessary for acidification to occur in a timely manner.
Acidification is a new management practice for western Oregon. See Acidifying Soil for Crop Production West of the Cascade Mountains (EM 8857-E) for more information.
Timing of acidification
If pH needs to be reduced by more than 0.2 unit:
begin the acidification process a year before planting. Acidification using elemental S relies on microorganisms for conversion of S to sulfate. This process requires warm, moist conditions. Even an early fall application does not provide sufficient time or soil warmth for the elemental S to completely react by the following spring (Figure 8).
Data for Figure 8 were collected from a silty clay or silty clay loam soil in the Willamette Valley (Table 1). A single 3,500 lb/a elemental S application incorporated in September did not decrease soil pH for 6 months. The initial decrease was slight, 0.2 to 0.3 pH unit. When the soil warmed in the summer, soil pH declined from 5.3 to 4.8 in approximately 4 months. The acidification reaction seemed to be complete a year after application, as soil pH decreased only 0.1 unit during the second winter after application.
| Figure 8 | Figure 9 | |
|---|---|---|
| Soil series | Not available | Malabon |
| Clay %* | 38.8 | 25 |
| Soil textural class | Clay loam/clay | Loam/clay loam |
| Cation exchange capacity | 34 meq/ 100 g soil | 21 meq/ 100 g soil |
| Initial soil pH | 5.6 | 6.4 |
| Initial S application date | September 20, 2003 | June 25, 2003 |
| Initial S application rate | 3,000 to 3,500 lb/a | 600 lb of 0-0-0-95/a |
| Second S application date | None | October 3, 2003 |
| Second S application rate | None | 600 lb of 0-0-0-95/a |
| Table in Acidifying Soil for Crop Production West of the Cascade Mountains used to estimate S rate | Table 7 | Table 5 |
*Determined by hydrometer method.
Figure 9 illustrates soil pH change from two elemental S applications, 3 months apart. Elemental S was initially applied in late June 2003 (Table 1). Three months later, soil pH had declined 0.5 pH unit, and the reaction was complete. A second application of elemental S was incorporated in October 2003. From October to March, soil pH declined only slightly. A sharp decline was measured after the summer of 2004. The reaction resulting from the second application was similar to that resulting from the fall application shown in Figure 8.
The data in Figures 8 and 9 highlight the need to acidify soil a year before planting. Be patient after a fall application, as the acidification process will begin slowly. Soil pH likely will decline only a few tenths of a unit until soils warm. In contrast, soil pH declines rapidly in the 3 to 4 months following a summer application.
The sites in Figures 8 and 9 were not irrigated. Winter and spring rainfall usually provides enough moisture for the acidification reaction.
No “best” time exists for application of elemental S; choose a time that fits your situation. In western Oregon, the soil usually is dry in the fall, making tillage and incorporation of S easier. Delaying S application until spring risks wet conditions for incorporation, but should not disrupt planting a year after incorporation if the soil is kept moist throughout the summer.
If initial soil pH is only 0.1 or 0.2 unit above the desired range:
Acidification and planting can be within a few months of each other. One option is to incorporate elemental S in the fall and plant in the spring, 4 to 6 months after S application.
If the soil pH is below 6.0 and container stock is planted, another option is to plant a few days after a spring or fall application of elemental S. If elemental S is incorporated shortly before planting, it will react after establishment.
Soil also can be acidified after establishment by adding an acid through the drip irrigation system. The resulting reduction in soil pH usually is 0.2 to 0.3 unit.
How much elemental S to apply
Soil differs dramatically in the S rate needed for acidification. The two soils in Figures 8 and 9 are excellent examples. Even though the initial soil pH in Figure 8 was 1 unit lower than that of the soil in Figure 9, it required three times the amount of elemental S. Not only were the S rates markedly different, so was the amount of acidification. The higher S rate applied to the soil in Figure 8 reduced soil pH 1 unit, while the lower rate in Figure 9 reduced soil pH 1.5 unit.
Elemental S rates vary for each field. The tables in Acidifying Soil for Crop Production West of the Cascade Mountains should be used only to estimate S rate. The tables are based on soil buffering capacity, or how easily soil pH can be changed. The primary agent buffering soil is cation exchange capacity (CEC). Soil analysis for CEC is a better guide than soil series for selecting the S rate table. Experience with your soil and situation is necessary to select an elemental S rate that will achieve your pH target.
Sawdust
Incorporation of Douglas-fir sawdust or compost prior to planting is a common industry practice. Recent research has shown that sawdust incorporated prior to planting may not improve plant growth in a good, well-drained sandy loam, loam, or silt loam. However, incorporation of an organic material such as sawdust can offer advantages in heavier soil.
To use sawdust preplant, apply 3.5 inches of sawdust in 3-foot-wide strips on 10-foot centers (19 units/acre). Add 5 lb N/unit of sawdust (95 lb N/acre) and incorporate to a depth of 10 inches. Sawdust contains little N. Soil microbes require N for sawdust decomposition, and they are more efficient at using soil N than are plants. Fertilizer N must be added so that normal microbial decomposition can occur without taking N from newly planted blueberries. Incorporated sawdust immobilizes more N than does sawdust applied to the surface.
Use of cedar, oak, walnut, or any other sawdust often is associated with chlorotic (yellow) leaves and poor growth. In addition, growers have lost a year or more of blueberry plant growth by incorporating horse bedding prior to planting; some horse bedding contains added lime and has a pH over 6.5. This material is not recommended for use in blueberry production.
The pH of Douglas-fir sawdust is about 5.5. Before using other organic material, test its pH.
Nitrogen (N)
N source
Blueberry plants use ammonium-N (NH4). Fertilizers that contain only nitrate (NO3), such as calcium nitrate, may cause injury or reduced growth and should be avoided. Ammonium nitrate is no longer sold as a fertilizer material because it can be used to make explosives. Calcium ammonium nitrate is a replacement product.
If the soil pH is below 5, do not use only ammonium sulfate (urea-sul) or ammonium phosphate-sulfate (16-20-0-15), as these materials may reduce pH too much. Use urea or a blend of urea with a small amount of ammonium sulfate.
Controlled-release fertilizers may be used provided the N source and the release rate are appropriate for plant needs. Note, however, that a single application of a more expensive product may not be more economical than multiple applications of a less expensive product. Oregon State University has not tested controlled-release N sources in producing blueberries.
Common N fertilizers are listed in Table 2.
| Fertilizer material | Chemical formula | Analysis or grade |
|---|---|---|
| Ammonium sulfate | (NHS4)2SO4 | 21-0-0 |
| Ammonium phosphate | NH4H2PO4 | 11-5-20 |
| Ammonium phosphate-sulfate | NH4H2PO4 (NH4)2SO4 | 16-20-0-15 |
| Calcium ammonium nitrate | CaCO3 + NH4NO3 | 27-0-0 |
| Calcium nitrate* | Ca(NO3)2 | 15.5-0-0 |
| Urea | CO(NH2)2 | 46-0-0 |
| Urea-sul | CO(NH2)2 + (NH4)2SO4 | 37 to 46% N + 4 to 8% S |
| Urea ammonium nitrate solution | CO(NH2)2 + NH4NO4 | 28 to 32% N |
* Not recommended for blueberries
Establishment (years 1 and 2)
Note: Year 1 is considered the first growing season, regardless of October or spring planting. Prune any flower buds present on nursery plants to prevent fruiting the first year. Pruning to prevent cropping in year 2 also is recommended in most situations. Allow only plants with sufficient vigor to produce a light crop in the second year. Early cropping (fruiting in years 1 and 2) has been shown to reduce root growth and yield in years 3 and 4.
Nitrogen application may not increase growth the first year. Most blueberry nursery stock contains sufficient N for the first year’s growth. Recent research showed no increase in whip length or plant dry weight from N application in the planting year. Even so, a small N application is prudent to protect the substantial investment involved in establishing blueberries.
We recommend applying 0.6 oz N per plant in year 1 and 0.7 oz N per plant in year 2. These rates assume that sawdust and 95 lb N/a were incorporated prior to planting and that 2 inches of surface mulch are used.
These rates are per plant. Because the root system may not yet have covered the entire in-row area, per-acre rates depend on plant spacing. Per-plant rates can be converted to per-acre rates as follows:
(oz/plant x plants/acre) ÷ 16 = lb N/acre
Remember to convert the N rate to the amount of product needed per acre (e.g., 100 lb N/a of ammonium sulfate [21-0-0] would be 100 ÷ 0.21 = 475 lb product/a).
Rabbiteye blueberries need much less N than do highbush blueberries and will have poor growth and yield with overfertilization. Grower experience has indicated that N rates can be reduced by up to half for rabbiteye blueberries with good results.
Time of application
Young and mature plants take up fertilizer N at the same time of year. Very little fertilizer (less than 2 percent) is taken up before the end of April. N should be “split,” with 0.2 oz (0.4 oz urea) applied in April, May, and June.
Controlled-release fertilizers may be used, provided the N source and the release rate are appropriate for plant needs. Note, however, that a single application of a more expensive product may not be more economical than multiple applications of a less expensive product. In experiments with a second-year planting, controlled release fertilizer provided no measurable advantage compared to ammonium sulfate. The only advantage was labor savings from a single application rather than several applications.
Application method
When plants are young and the canopy does not fill the row, we recommend applying fertilizer by hand. Spread the recommended rate in an area under the drip line of the bush (Figure 10). Apply fertilizer evenly.
Use caution when applying N to young blueberries, as over-application will cause leaf burn, and severe over-application will cause plant death. Young plants are much more sensitive to under- and over-fertilization because they do not have a lot of stored N reserves and because the young root system is very sensitive to fertilizer “burn.”
Applying all of the N fertilizer in a concentrated area near the crown can cause plant death.
If fertilization must be done by machine, you will need to increase the rate because plants cannot take up fertilizer that is applied beyond the rooting area. You must calculate how much of the application is within the drip line of the bush and how much is “wasted” and adjust the application rate accordingly.
Producing plants (starting in year 3)
Adequate tissue N levels are necessary for renewal growth, crop production, and flower bud development for next year's crop. Excess N early in the season may reduce fruit quality, increase risk of disease on fruit, and delay maturation. Excess N late in the season leads to excessive vegetative growth, a late flush of growth (which increases risk of winter injury), and reduced flower bud development.
In general, fertilizer rates should increases as the planting ages (Table 3). Rates for years 3 and 4 are per-plant, as the root system may not yet cover the entire in-row area, thus, per-acre rates would depend on plant spacing. Once plants "fill the row", or their canopies touch, per-acre rates no longer are affected by plant spacing.
Remember to convert the N rate to the amount of product needed per acre (e.g., 100 lb N/a of ammonium sulfate [21-0-0] would be 100 ÷ 0.21 = 475 lb product/acre).
The rates in Table 3 assume that sawdust and 95 lb N/a were incorporated prior to planting. These rates are for fields with a surface Douglas-fir sawdust mulch (typical carbon-to-nitrogen ratio of 800:1) replenished every few years. Blueberries topdressed with other kinds of mulch with a higher C:N ratio, or to a greater depth, may need more fertilizer N. For example, if you apply an additional 11 units of sawdust (2 inches deep in 3-foot-wide bands on 10-foot centers), you will need to add 25 lb N/a to compensate for N immobilized by the fresh sawdust. In contrast, reduce the N fertilizer rate if no surface organic mulch is used.
| Year | N (oz/plant) | N (lb/a)* |
|---|---|---|
| 3 | 0.8 | - |
| 4 | 0.9 | - |
| 5 | - | 100 |
| 6 | - | 125 |
| 7 | - | 145 |
| 8+ | - | 165 |
| Leaf N (%) | Status |
|---|---|
| below 1.50 | Defficient |
| 1.50 - 1.75 | Below Normal |
| 1.76 - 2.00 | Normal |
| 2.01 - 2.05 | Above Normal |
| above 2.50 | Excess |
Once the planting reaches maturity (year 7 or 8), apply 145 to 165 lb N/a. However, adjust N rates, if necessary, based on observations of growth, tissue N analysis (Table 4), and the amount and type of mulching.
Soil testing for N is not a reliable indicator of perennial crop N status. Leaf analysis alone (Table 4) does not indicate whether N fertilization is required, but it can be used in conjunction with an assessment of plant growth and productivity to determine N status (see “Interpreting tissue test results,” page 3).
Keep in mind that as sawdust ages and decomposes, it will release some N that is available to the plants. We do not know how quickly this process occurs. Thus, keep monitoring plant growth and N tissue concentrations. Before using any other organic material, test its pH.
The N rates in this publication take into account “efficiency” of uptake. Plants cannot take up all of the fertilizer applied, regardless of the product or method of application. Efficiency of uptake is affected by plant age (depth and width of root zone), coverage of the in-row area (uptake efficiency increases as plants fill the row), method of application (granular, liquid, or foliar applications), and the amount and timing of application. Rabbiteye blueberries need much less N than do highbush blueberries and will have poor growth and yield with overfertilization. Grower experience has indicated that N rates can be reduced by up to half for rabbiteye blueberries with good results.
Time of application
Young and mature plants take up fertilizer N at the same time of year. Very little fertilizer (less than 2 percent) is taken up before the end of April and less than 10 percent after mid-July. The blueberry plant relies on stored N reserves early in the season. A relatively clear “application window” for efficient N fertilizer uptake exists between late April and mid-June.
The total N fertilizer rate should be split into thirds, with the first application in late April, the second in midMay, and the third in mid-June. Blueberries are shallow rooted and inefficient at using fertilizer N. Multiple applications provide the N fertilizer when plants need it and allow plants more time to absorb N before it is moved below the root zone.
Application method
When plants are young and the canopy does not fill the row, we recommend fertilization by hand. Spread the recommended rate in an area under the drip line of the bush. If fertilization must be done by machine, increase the rate, as any fertilizer not above the roots will be unavailable to the plants. Once the canopy is full and plants "touch," fertilizer uptake efficiency is much greater.
The least expensive method of fertilizer application is to apply a granular product as a broadcast band in the row. Recommended per-acre rates in this publication are based on this method of application.
Little lateral nutrient movement occurs within the plant. Thus, fertilizer (whether applied as a granular material or via drip irrigation) should be as evenly distributed under the plant canopy as possible to ensure even growth. Any N fertilizer application should be followed by rain or irrigation as soon as possible.
Foliar application is an inefficient method of applying N fertilizer to blueberry plants. No more than 5 percent urea (in water), the equivalent of about 14 lb N/a, can be applied to plants without burning leaves. However, not all of this N is taken up by plants. In research with other crops, no more than 50 percent (or 7 lb N/a) of foliarapplied N was taken up. Because of their waxy cuticle, blueberry leaves are not very effective at taking up N. Most foliar-applied N that is taken up by plants is taken up through the roots (runoff).
Phosphorus (P)
Symptoms of P deficiency—stunted plants with dark, purplish-green, small leaves—rarely are seen in Oregon. Pears planted in Parkdale soil in the Hood River area are the only documented case of P deficiency in deciduous fruit in western Oregon. Leaves sometimes have a purple color in early spring when temperatures are cool and soils are wet—a sign of temporary P deficiency. This condition will be corrected when soils warm. Effects from excessive P also are rare.
P application might be justified if tissue P and soil P levels are low. Soil test P alone is a poor indicator of P sufficiency for blueberries.
No universal tissue concentration is used to define blueberry P deficiency. Michigan Extension publications define P deficiency as below 0.07 percent P, Massachusetts below 0.05 percent, and Wisconsin and Minnesota below 0.08 percent. Pennsylvania, Massachusetts, Wisconsin, and Minnesota define optimum P as above 0.1 percent, the value used in this publication.
Apply P according to Table 5, based on tissue concentration. These rates are appropriate for any plant spacing. Keep in mind that P is not mobile in the soil; thus, changes in tissue P occur a year or two after application.
Apply P in late fall or spring prior to budbreak. When N is included with P and K, apply in spring. Irrigate after application if rain does not fall within 1 or 2 days.
Various fertilizers can be used: triple superphosphate (0-45-0), ammonium phosphate (11-52-0), or ammonium phosphate sulfate (16-20-0-15).
| Soil test P (Bray) (ppm) | Leaf P (%) | Apply this amount of P2O5 (lb/a) |
|---|---|---|
| 0 - 25 | below 0.07 | 40 - 60 |
| 26 - 50 | 0.08 - 0.10 | 0 - 40 |
| above 50 | above 0.10 | 0 |
| Soil test K (ppm) | Tissue K (%) | Apply this amount of K2O (lb/a) |
|---|---|---|
| 0 - 100 | below 0.20 | 75 - 100 |
| 101 - 150 | 0.21 - 0.40 | 0 - 75 |
| above 150 | above 0.40 | 0 |
Potassium (K)
Potassium deficiency is relatively rare in Oregon. Leaves of K-deficient plants may develop several symptoms, including scorching along the margin, cupping, curling, and necrotic spots. Low leaf K values may be caused by poor drainage, drought, or very acid soils.
Yield also has a strong influence on leaf K levels. Potassium concentration in the berry increases dramatically as fruit matures, averaging more than 60 mg per berry when ripe. Thus, deficiency levels in leaves may occur in a heavy yield year, with normal levels returning after harvest.
Normal August leaf K levels are 0.41 to 0.70 percent. Blueberry yields have been shown to increase with K fertilization on various soil types if K is deficient. If a below-normal leaf value occurs, band trial applications of 150 lb/a potassium sulfate (0-0-52) [K2SO4]. Use Table 6 as a guide, keeping in mind that application rates for established plantings should be based on tissue concentration. Use these rates regardless of plant density.
Avoid muriate of potash (potassium chloride), because chloride may cause reduced growth or injury to blueberries. If both K and Mg are needed, use 400 lb/a sulfate of potash-magnesia (0-0-21, 11% S, 10% Mg) [K2SO4, MgSO4]. Excess K can interfere with uptake of other elements, especially Mg. Therefore, do not apply K unless foliar analysis indicates a deficiency.
Apply K in late fall or in spring prior to budbreak. When N is included with P and K, apply in spring. Irrigate afterward if rain does not fall within 1 or 2 days.
Potassium is immobile in soil. Thus, changes in blueberry tissue K occur a year or two after application.
pH and manganese (Mn)
pH and manganese (Mn) The critical soil pH range for blueberries is 4.5–5.5. If the soil pH is above this range, use elemental sulfur to reduce soil pH before planting. (See “Site preparation,” page 4, and Acidifying Soil for Crop Production West of the Cascade Mountains). Soil pH will begin to decrease within 4 to 5 months if soil moisture and temperature are adequate for growth. Before and after planting, sample again to determine whether further acidification is necessary.
Keep in mind that use of ammonium sulfate fertilizer as a source of N will lower soil pH. If soil pH declines in the surface 6 inches or top foot of soil, increasing pH is extremely difficult and often impossible since lime is not mobile in soil. Thus, application of a small amount of lime when soil pH drops below 5.0 is prudent. To maintain soil pH above 4.5, apply 200 to 300 lb lime/a annually in the fall. If the soil pH is below 4.0, apply 1 to 2 ton lime/a. Sample and test soil pH in the surface 3 inches of soil 8 months after lime application. Do not apply lime in spring before N fertilizer is used.
Foliar manganese concentration normally is between 30 and 350 ppm. Tissue Mn increases as soil pH decreases; thus, tissue Mn can serve as a guide to soil pH changes. Blueberry tissue Mn above 450 ppm is considered excessive and an indictor of decreasing soil pH and the need for lime application. Blueberry plants grow normally with Mn concentration as high as 650 ppm. Vaccinium plants (blueberries and relatives) are thought to have a mechanism for Mn tolerance, since Mn levels can be as high as 4,000 ppm in lowbush blueberries without affecting plant growth. Even so, sustained tissue Mn concentration of 500 ppm or above is not recommended.
In addition to low soil pH, fungicide and foliar micronutrient application can cause high (above 450 ppm) tissue Mn results. These materials are “coatings” on leaves and not a measure of Mn in leaf tissue. Before beginning a liming program based on high tissue Mn, confirm that no foliar Mn application has been made and check soil pH.
When soil sampling an established blueberry planting, keep in mind that soil pH differences will occur throughout the field. Close to the plants, where N fertilizers are applied, the pH will be lower than between rows (Figure 5, page 4).
Calcium (Ca)
Normal blueberry tissue Ca concentration is from 0.41 to 0.8 percent. Although blueberries seldom are deficient in Ca, variations in leaf Ca occur. High leaf Ca concentration might indicate high soil Ca or a high crop load. Low leaf Ca concentration can be present in heavily fertilized, vigorously growing plants, or it might be a result of low soil pH. If low soil pH is suspected, check the tissue Mn concentration. Abnormally high Mn (above 450 ppm) is an indication of low soil pH and low soil Ca.
If Ca is required and pH is below 4.0, topdress 0.5 to 1 ton/a agricultural limestone (Ca). If Mg is needed as well, substitute dolomitic limestone (Ca + Mg). If Ca is needed and the pH is above 5.0, apply 0.5 to 1 ton gypsum/a. Apply these materials in the fall to permit time for reaction with soil before the growing season.
Sulfur (S)
Sulfur is an essential element. However, it is more commonly applied to blueberries to reduce soil pH than to correct a nutrient deficiency. Normal leaf levels for S range from 0.11 to 0.16 percent.
Boron (B)
Boron deficiency, which is relatively common in Oregon, causes tip dieback. Leaves close to aborted shoot tips develop a mottled chlorosis and cupped shape. Leaf and fruit buds fail to develop in severely deficient plants. Winter injury also may be greater on B-deficient plants.
Use Table 7 as a guide to fertilization. If B is deficient, apply either Solubor (20% B) at 2 to 6 lb product/l00 gal water/a, or 10 to 20 lb/a borax (11% B).
Apply Solubor or an equivalent material as a foliar spray before bloom or after harvest and before leaf senescence. Apply borox in the fall or early spring prior to rain.
| Leaf B (ppm) | Status |
|---|---|
| below 20 | Deficient |
| 21 - 30 | Below normal |
| 31 - 80 | Normal |
| 81 - 150 | Above normal |
| above 150 | Excess |
Many blueberry growers in Oregon follow an annual B application program of 0.5 lb B/a. Monitor leaf B levels carefully. Excess B is toxic to plants.
Zinc (Zn)
Zn deficiencies are less common than B deficiencies in Oregon. Symptoms of Zn deficiency include short internodes and small leaves. The youngest leaves are somewhat yellow and folded upward along the midrib. Excessive use of P sometimes results in Zn deficiency symptoms. Zn deficiency is less common when pH is below 6.0.
Normal leaf concentrations are from 8 to 30 ppm. If plants are deficient, apply one fo the following:
- Zn chelate (14% Zn) as a foliar spray at 1 lb/100 gal water/a after harvest
- 10 to 30 lb/a Zn chelate or 10 to 30 lb/a Zn sulfate (36% Zn) to the soil
Copper (Cu)
Copper deficiency is rare in Oregon blueberries. Symptoms of Cu deficiency include yellowing between veins of young leaves and, in severe cases, young shoot dieback. Cu deficiency may be more severe on soils with more than 25 percent organic matter.
Normal leaf levels for Cu are from 5 to 15 ppm. If tissue concentration is below 3 ppm, use a trial application of copper sulfate (25% Cu). Broadcast at 30 to 50 lb/a or apply 1 lb/100 gal water/a as a foliar spray anytime leaves are present. Cu is very toxic at an excessive rate, so use caution and experiment on a few plants before applying to the entire field.
Iron (Fe)
Iron deficiency in leaves often is the result of high soil pH rather than Fe deficiency in the soil. Symptoms of Fe deficiency include yellowing between leaf veins, appearing first on the younger leaves. Shoot growth and leaf size sometimes are reduced.
Normal Fe leaf values are 61 to 200 ppm. If leaf levels are deficient, apply iron chelate (10% Fe) at 2 lb/100 gal water/a twice as a foliar spray when leaves are present (see label) or 15 to 30 lb/a to the soil. A soil application of 10 to 20 lb/a ferrous sulfate (34% Fe) also is suitable.
For more information
OSU Extension publications
- Acidifying Soil for Crop Production West of the Cascade Mountains (Western Oregon and Washington), EM 8857-E, by D. Horneck, J. Hart, R. Stevens, S. Petrie, and J. Altland (2004).
- Blueberry Economics: The Costs of Establishing and Producing Blueberries in the Willamette Valley, EM 8526-E, by B. Eleveld, B. Strik, K. DeVries, and W. Yang (2005).
- Fertilizer and Lime Materials, FG 52, by J. Hart (1992). Highbush Blueberry Production, PNW 215, by B. Strik et al. (1993).
- Laboratories Serving Oregon: Soil, Water, Plant Tissue, and Feed Analysis, EM 8677, by J. Hart (2006).
- Monitoring Soil Nutrients Using a Management Unit Approach, PNW 570-E, by M. Staben, J. Ellsworth, D. Sullivan, D. Horneck, B. Brown, and R. Stevens (2003).
- Soil Sampling for Home Gardens and Small Acreages, EC 628, by M. Robotham and J. Hart (2003).
Other publications
- Ballinger, W.E. 1966. Seasonal Trends in Wolcott Blueberry (Vaccinium corymbosum L.) Leaf and Berry Composition. North Carolina Agricultural Experiment Station Technical Bulletin 173.
- Ballinger, W.E. and L.J. Kushman. 1966. Factors affecting the mineral element content of leaves and fruit of Wolcott blueberries. Proc. Amer. Soc. Hort. Sci. 88:325–330.
- Bañados, M.P. 2006. Dry Weight and 15N-nitrogen Partitioning, Growth, and Development of Young and Mature Blueberry Plants. Oregon State University Department of Horticulture, Ph.D. dissertation.
- Eck, P. 1988. Blueberry Science. Rutgers University Press. 284 pp.
- Gough, R.E. 1984. Split root fertilizer application to highbush blueberry plants. HortScience 19:415–416.
- Harmer, P.M. 1944. Soil reaction and growth of blueberry. Soil Sci. 9:133–144.
- Martin, L.W. and P.J. Pelofske. 1983. Ammonium sulfate fertilization of blueberries on a mineral soil. Commun. in Soil. Sci. Plant Anal. 14:131–142.
- Proceedings for the 2004 Blueberry Production Course, Salem, OR. Oregon State University Department of Horticulture.
- Retamales, J.B. and E.J. Hanson. 1989. Fate of 15N-labeled urea applied to mature highbush blueberries. J. Amer. Soc. Hort. Sci. 114:920–923.
- White, L. 2006. The Effect of Pre-plant Incorporation with Sawdust, Sawdust Mulch, and Nitrogen Fertilizer Rate on Soil Properties and Nitrogen Uptake and Growth of ‘Elliott’ Highbush Blueberry. Oregon State University Department of Horticulture, M.S. thesis.
- Woodbridge, C.G. and R.H. Drew. 1960. A boron deficiency die-back in highbush blueberry. Plant Disease Reporter 4A:855–857.