Eyespot of wheat, also known as strawbreaker foot rot, is a lower stem-infecting foot rot pathogen that limits winter wheat yields in the dryland Pacific Northwest.
The scientific nomenclature of eyespot has undergone many changes, which can lead to confusion for the common name of the disease.
For the purpose of this publication we will refer to Oculimacula yallundae and Oculimacula acuformis as the causal agents of eyespot of wheat. Both species can co-exist within the same field, cause identical symptoms on wheat, and follow the same disease cycle. Management recommendations for both species are interchangeable.
Eyespot, or strawbreaker foot rot: A disease by many other names
Previously known as:
- Fungus name:
- Cercosporella herpotrichoides
- Pseudocercosporella herpotrichoides
- Disease name:
- Cercosporella foot rot
- Pseudocercosporella foot rot
Two causal agents:
- Current scientific names:
- Oculimacula yallundae
- Oculimacula acuformis
- Obsolete scientific names:
- P. herpotrichoides var. herpotrichoides
- P. herpotrichoides var. acuformis
- Tapesia acuformis
- Tapesia yallundae
What eyespot is not: Rhizoctonia cerealis, the causal agent of sharp eyespot of wheat.
Disease cycle
Eyespot survives on infected wheat straw residue between seasons until autumn rains spread inoculum to autumn-sown seedlings. The infection can begin at the coleoptile and continues through fall, winter, and early spring. Conidia, or asexual spores, germinate from previously infected crop residue and directly penetrate coleoptiles and leaf sheaths at the ground surface. Wind-borne infection has been documented in Australia, Germany, and the United Kingdom. However, infection is more likely to be spread by rain.
Depending on environmental conditions, symptoms may not be visible until spring. Infection results in characteristic elliptical or eye-shaped lesions on the lower stem (Figure 2A). Lesions typically have a dark brown perimeter that distinguishes between healthy and infected tissue (Figure 2B). The center of each lesion is typically yellowish-brown and may have dark brown-blackish pseudoparenchyma fruiting bodies (Figure 2B). In light infections, eyespot symptoms may be limited to leaf sheath layers with no progression into the culm. In severe infections, the lesion will penetrate the stem tissue, and a whitish-gray mass of fungus, called mycelium, may be visible inside the hollow wheat stem (Figure 2C). Eyespot lesions are brittle in contrast to surrounding healthy tissue and thus weaken the stem (Figure 1), which may result in multidirectional lodging (Figure 3). Lesions can also girdle the flow of water and nutrients to the ripening wheat head, resulting in prematurely ripened white heads that are visible after heading but before the crop dries down prior to harvest (Figure 3).
Yield loss
Yield loss depends upon the degree and timing of the disease and the extent of lodging. Losses of up to 50 percent in winter wheat fields with uniform eyespot infection have been documented. Superficial eyespot infections that do not fully penetrate the culm typically do not affect the flow of water or nutrients through the plants, or result in lodging or significant yield loss.
Eyespot infection of spring wheat is possible but of minor economic importance.
Once eyespot is established in a field, it can remain indefinitely. Regions with limited rotations of winter wheat with cool, moist, fall seeding conditions, such as the dryland Pacific Northwest, are particularly vulnerable to eyespot infection. Spores can overwinter in the stubble of the previous year’s crop, perpetuating the cycle. Factors that may increase risk of eyespot infection include:
- Planting a susceptible variety
- Past history of eyespot in the field leading to inoculum reserves
- Early planted wheat, leading to increased production of fall biomass
Eyespot is mainly a dryland wheat issue, although it can occur in irrigated wheat and in regions that receive a lot of rain. Greater moisture availability contributes to straw decomposition, which reduces inoculum much more quickly. Growing wheat in a short rotation in wet conditions increases the chance of eyespot infection.
Genetic resistance
Planting genetically resistant varieties is the most effective and economically viable way to prevent yield loss from eyespot.
Two named genes confer a level of resistance to eyespot in wheat: Pch1 and Pch2. Pch1 was derived from a wild grass species, and Pch2 was derived from the popular European wheat variety ‘Cappelle Desprez’. Both genes confer resistance to both O. yallundae and O. acuformis.
The resistance gene Pch1 provides a greater degree of resistance than Pch2. The winter wheat cultivar ‘Madsen’ was the first eyespot-resistant cultivar in the United States containing Pch1, though many more have subsequently been released (Table 1).
Table 1. Pacific Northwest winter wheat varieties containing the Pch1 or Pch2 genes
for resistance to wheat eyespot
| Variety | Market class | Pch1 | Pch2 | Clearfield | Source |
|---|---|---|---|---|---|
| Bobtail | Soft white | No | Yes | No | Oregon State University |
| Nixon* | Soft white | Yes | No | No | Oregon State University |
| Rosalyn | Soft white | Yes | Yes | No | Oregon State University |
| Tubbs 06 | Soft white | Yes | No | No | Oregon State University |
| Weatherford | Soft white | Yes | No | No | Oregon State University |
| ORCF-102 | Soft white | Yes | No | 1-gene | Oregon State University |
| OR2×2 CL+** | Soft white | Yes | Yes | 2-gene | Oregon State University |
| ARS-Selbu | Soft white | Yes | Not determined | No | USDA Agricultural Research Service/Washington State University |
| Finch | Soft white | Yes | Not determined | No | USDA Agricultural Research Service/Washington State University |
| Madsen | Soft white | Yes | No | No | USDA Agricultural Research Service/Washington State University |
| Masami | Soft white | Yes | Not determined | No | Washington State University |
| Otto | Soft white | Yes | Not determined | No | Washington State University |
| Puma | Soft white | Yes | Not determined | No | Washington State University |
| Purl | Soft white | Yes | Not determined | No | Washington State University |
| LCS Sonic | Soft white | Yes | Not determined | No | Limagrain Cereals Seeds |
| SY Banks | Soft white | Yes | Not determined | No | AgriPro |
| SY Candor | Soft white | Yes | Not determined | No | Agripro |
| ARS-Chrystal | Club | Yes | Not determined | No | USDA Agricultural Research Service/Washington State University |
| ARS-Crescent | Club | Yes | Not determined | No | USDA Agricultural Research Service/Washington State University |
| Cara | Club | Yes | No | No | USDA Agricultural Research Service/Washington State University |
| Chukar | Club | Yes | Not determined | No | USDA Agricultural Research Service/Washington State University |
| Coda | Club | Yes | No | No | USDA Agricultural Research Service/Washington State University |
| Hyak | Club | Yes | Not determined | No | USDA Agricultural Research Service/Washington State University |
| Pritchett | Club | Yes | Not determined | No | USDA Agricultural Research Service/Washington State University |
| LCS Colonia | Hard red | Yes | Not determined | No | Limagrain Cereals Seeds |
** Experimental No. ORI2150031 CL+
Pch2 is less characterized in Pacific Northwest germplasm, but the cultivar ‘Bobtail’ is known to have Pch2, but not Pch1.
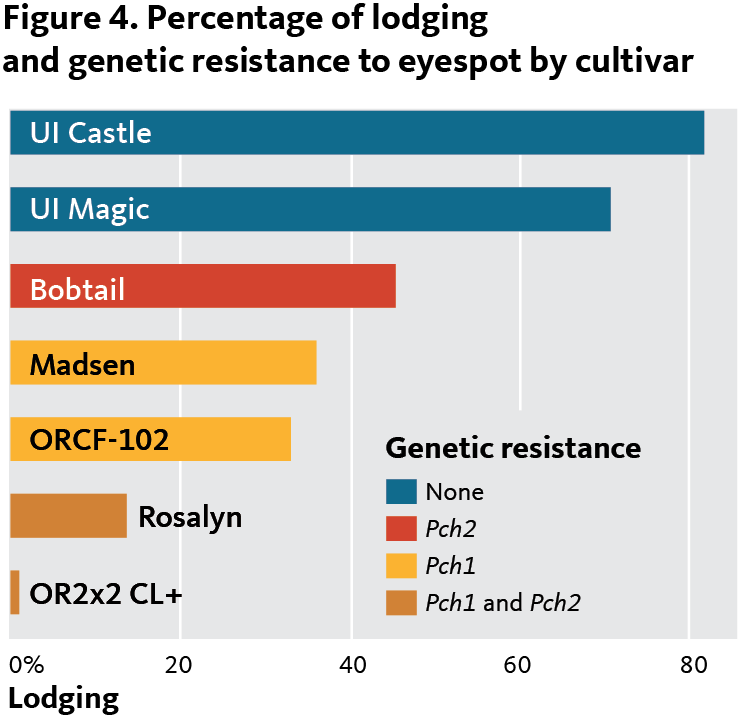
Figure 4 shows the degree of lodging caused by eyespot in inoculated field plots for two highly susceptible cultivars; two cultivars with Pch1; ‘Bobtail’ with Pch2 only; and two cultivars combining Pch1 and Pch2. Combining the two resistance genes appears to provide a significant positive synergism for increased resistance.
The real world is more complex than the graph in Figure 4, however; many resistance genes of small effect have not yet been well characterized. As a result, cultivars vary significantly in their resistance to eyespot, beyond the resistance conferred by Pch1 and Pch2.
Because of this variation, Oregon State University screens a collection of elite winter wheat varieties annually in inoculated trials in Corvallis. The trial aims to identify resistant cultivars and inform recommendations to growers. Inoculated plots are rated for lodging on a quantitative scale for eyespot resistance. Table 2 shows ratings for current cultivars that have been evaluated in at least two recent growing seasons.
Table 2. Eyespot resistance rating based on lodging in inoculated field plots.
For current cultivars that have been evaluated in at least two growing seasons in recent years.
1= highly resistant, 9 = highly susceptible
Soft white, non-Clearfield:
- Bobtail: 6
- Jasper: 9
- Kaseberg: 9
- LCS Art Deco: 8
- LCS Biancor: 8
- LCS Drive: 7
- LCS Shark: 8
- Mary: 7
- Nixon: 6
- Norwest Duet: 9
- Norwest Tandem: 7
- Rosalyn*: 3
- Stephens: 7
- SY Assure: 8
- SY Banks: 4
- SY Dayton: 4
- SY Ovation: 7
- Tubbs 06: 5
- WB1529: 8
Hard white, non-Clearfield:
- Irv 6 Whetstone: 8
Soft white, Clearfield:
- Curiosity CL+: 9
- Mela CL+: 9
- LCS Biancor: 8
- LCS Art Deco: 8
- 8 ORCF-101: 7
- ORCF-102: 4
- OR2×2 CL+*: 2
- UI Castle CL+: 9
- UI Magic CL+: 9
- UI Palouse: 4
- WB1376 CL+: 4
Hard red, non-Clearfield:
- Keldin: 8
- LCS Azimut: 5
- LCS Aymeric: 4
- LCS Colonia: 4
- LCS Evina: 4
- LCS Jet: 4
- Norwest 553: 6
- SY Touchstone: 4
- WB Arrowhead: 8
- Whetstone: 8
Hard red, Clearfield:
- SY Clearstone CL2: 9
- WB4623 CLP: 8
* Cultivars that carry both Pch1 and Pch2
Fungicides
Timing
When an eyespot-susceptible cultivar is planted, fungicides may be used to control infection. Apply fungicide before stem elongation for maximum efficacy. Timing depends on regional factors and is heavily influenced by weather.
In the dryland Pacific Northwest, fungicides are often tank mixed with the late winter herbicide application, commonly referred to as “herbicide timing” application. Unfortunately, in most typical years, an herbicide timing fungicide application to control eyespot will wear off before the jointing phase is complete, leaving the plant unprotected against additional infections.
Applying fungicide after jointing will not be effective, because eyespot damage has already been initiated. A fungicide application after lodging would not prevent yield loss.
Scout and sample to determine whether an eyespot infection is severe enough to warrant fungicide.
To scout for eyespot spray threshold, sample at least 10 plants (totaling at least 50 tillers) from representative areas throughout the field. Wash or soak the samples in water to remove excess soil. Examine the washed tillers for characteristic eyespot lesions—outer leaf sheaths will have a brownish appearance (Figure 1). When leaf sheaths are stripped away, an elliptical lesion will be revealed on the stem (Figures 2A, 2B). If 10 percent of the collected tillers have obvious eyespot lesions in the period from early March to mid-April, consider applying fungicide.
Global resistance issues
Due to the lack of genetic resistance to eyespot available until the 1980s, wheat production in many parts of the world, including the United States, relied on benzimidadole (Group 1, MBC) fungicide sprays such as benomyl, carbendazim, or thiophanate-methyl for eyespot control. Eyespot is classified as a medium risk pathogen for developing fungicide resistance. Eyespot resistance to MBC (Group 1) fungicides is likely because MBC fungicides are high risk for developing fungicide resistance, MBC resistance is common in many other fungal species, and resistance is conferred by several target-site mutations.
The UK’s Agricultural Development and Advisor Service estimated that 52 percent of winter wheat crops were sprayed one or more times with an MBC fungicide in 1982 for eyespot control. Benomyl resistant eyespot was first detected in Germany in 1975, and was detected in the UK in 1981 after two growers in the UK reported severe eyespot infections in fields that had previously received a timely application of an MBC fungicide. This discovery prompted a larger survey of the UK for MBC-resistant eyespot in 1982. Results of this survey detected MBC resistance in 40 percent of surveyed fields.
Local resistance issues
More than 1.2 million acres of winter wheat across Oregon, Washington, and Idaho were affected by eyespot prior to the release of genetically resistant cultivars adapted to the Pacific Northwest. The majority of affected acreage received a benzimidazole fungicide spray annually for eyespot control, which put heavy selection pressure on eyespot to favor the buildup of MBC-resistant eyespot isolates. This caused a fungicide resistance issue similar to that of the UK to unfold in the 1980s in the Pacific Northwest, with the detection of benzimidazole-resistant eyespot isolates in commercial winter wheat fields
There is positive cross-resistance between MBC group members, meaning fungal resistance to a benomyl product confers resistance to all other MBC group members, including carbendazim, fuberidazole, thiabendazole, thiophanate, and thiophanate-methyl.
Benomyl was federally registered for use on wheat for eyespot control in 1977, and a spring benzimidazole fungicide application before stem elongation became standard practice in the dryland Pacific Northwest for eyespot control. At the time of the MBC-resistant eyespot discovery in the Pacific Northwest in 1989, the majority (ranging from 67 to 100 percent) of isolates sampled from nine commercial winter wheat fields were resistant to benzimidazole. Despite the discovery of MBC-resistant eyespot in the Columbia Basin by Murray in 1996, many growers in the region still apply MBC products for eyespot control; however, these applications have little to no effect in controlling eyespot.
Fungicide alternatives
Strobilurin blends registered in Oregon, though effective against stripe rust, are not effective in controlling eyespot. The DMI fungicide prothioconazole, alone or in mixtures, has shown promise in Washington, and is considered a good fungicide for eyespot control in the UK. Priaxor, a blend of the succinate dehydrogenase inhibitor fluxapyroxad and the strobilurin pyraclostrobin, has shown better eyespot control than several other fungicides tested in Washington.
Due to the expense of these fungicides and the risk of fungicide resistance, scout-based spray programs are key to achieve economic returns on spray investment.
2017 MBC resistance case study
In June 2017, a producer in Adams, 10 miles north of the Columbia Basin Agricultural Research Center, reported a severe eyespot infection. The 40 foot-by-100 foot winter wheat drill strip variety trial with an eyespot-susceptible cultivar was nearly 100 percent lodged (Figure 4).
Recommended scouting procedures were followed, and 92 percent of sampled tillers showed characteristic eyespot lesions. The field had a history of eyespot and received a timely application of Topsin-M (thiophanate-methyl, UPI) before stem elongation. Isolations were made from infected stems using a protocol modified from Murray and molecularly confirmed as O. yallundae. Evidence showed the addition of thiophanate-methyl did not significantly reduce O. yallundae growth after eight days of incubation (p=0.73), 12 days of incubation (p=0.54), or 35 days of incubation (p=0.27). In contrast, strong evidence showed the addition of thiophanate-methyl significantly reduced growth of O. acuformis and O. yallundae 1990 reference isolates at all concentrations tested, and at all incubation lengths (p<0.001).
Conclusion
Eyespot of wheat has a regional distribution in the dryland Pacific Northwest and can cause significant yield loss if conditions are favorable. The most economical method for avoiding yield loss due to eyespot is host genetic resistance. If not planting a resistant cultivar, deploy a scout-based fungicide spray program to limit economic loss.
References
- Bateman, G.L., and Taylor, G.S. 1976. Significance of the coleoptile in the establishment of seeding infection on wheat by Pseudocercosporella herpotrichoides. Transactions of the British Mycological Society 67.
- Bockus, W.W., Bowden, R.L., Hunger, R.M., Morrill, W.L., Murray, T.D., and Smiley, R.W. 2010. Compendium of Wheat Diseases and Pests, Third Edition, American Phytopathological Society Press.
- Coyier, D.L., Herman, D., Eckert, J. W., Gaede, H., Gregory, G., Hickey, K. D., et al. 1985. The biologic and economic assessment of Benomyl—A report of the Benomyl assessment team to the rebuttable presumption against registration of Benomyl. Accessed Oct. 9, 2018.
- Deighton, F.C. 1973. Studies on Cercospora and allied genera. IV. Cercosporella Sacc., Pseudocercosporella gen. nov. and Pseudocercosporidium gen. nov. Mycological Papers. Accessed Oct. 2, 2018.
- Doussinault, G., Delibes, A., Sanchez-Monge, R., and Garcia-Olmedo, F. 1983. Transfer of a dominant gene for resistance to eyespot disease from a wild grass to hexaploid wheat. Nature. 303:698–700.
- Dyer, P., Furneaux, P., Douhan, G., and Murray, T. 2001. A multiplex PCR test for determination of mating type applied to the plant pathogens Tapesia yallundae and Tapesia acuformis. Fungal Genetics and Biology. 33:173–180.
- FRAC. 2005. FRAC code list. Accessed April 2, 2015.
- FRAC. 2013. Pathogen risk list.
- Fron, G. 1912. Contributiona l’étude de la maladie de ‘pied noir des cereales’ ou ‘maladie du piétin.’ Annales de la Science Agronomique Française et Étrangere, série. 4:3–29.
- Gosling, P. 2018. Fungicide activity and performance in wheat. Agriculture and Horticulture
- Development Board. Warwickshire, UK. Accessed Feb. 14, 2019
- Griffin, M.J., and Yarham, D.J. 1983. Fungicide resistance—MBC resistance in the eyespot fungus. Agrospray:2–5.
- Hollins, T. W., Lockley, K.D., Blackman, J.A., Scott, P.R., and Bingham, J. 1988. Field performance of Rendezvous, a wheat cultivar with resistance to eyespot (Pseudocercosporella herpotrichoides) derived from Aegilops ventricosa. Plant Pathology. 37:251–260.
- Klos, K.L.E., Wetzel, H.C., and Murray, T. D. 2014. Resistance to Oculimacula yallundae and Oculimacula acuformis is conferred by Pch2 in wheat. Plant Pathology. 63:400–404.
- Leonard, J.M., Watson, C.J. W., Carter, A.H., Hansen, J.L., Zemetra, R.S., Santra, D.K., et al. 2008. Identification of a candidate gene for the wheat endopeptidase Ep-D1 locus and two other STS markers linked to the eyespot resistance gene Pch1. Theoretical and Applied Genetics. 116:261–270.
- Lewien, M.J., Murray, T.D., Jernigan, K.L., Garland-Campbell, K.A., and Carter, A.H. 2018. Genome-wide association mapping for eyespot disease in U.S. Pacific Northwest winter wheat. PloS One. 13:e0194698.
- Lucas, J.A., Dyer, P.S., and Murray, T.D. 2000. Pathogenicity, host specificity, and population biology of Tapesia spp., causal agents of eyespot disease of cereals. Advances in Botanical Research. 33:225-258.
- Murray, T.D. 1996. Resistance to Benzimidazole fungicides in the cereal eyespot pathogen, Pseudocercosporella herpotrichoides, in the Pacific Northwest 1984 to 1990. Plant Disease. 80:19–23.
- Murray, T.D. 2006. Strawbreaker foot rot or eyespot of wheat. Washington State University Extension. EB1378:1–6.
- Murray, T.D., Parry, D.W., Cattlin, N.D., Parry, D.W., and Cattlin, N.D. 2008. Diseases of small grain cereal crops: a colour handbook. CRC Press. Accessed Aug. 29, 2018.
- Nirenberg, H. 1981. Pseudocercosporella herpotrichoides var. acuformis. Zeitschrift für Pflanzenkrankheiten und Pflanzenschutz. MB 117692:241–248.
- Rashid, T., and Schlosser, E. 1975. Resistenz von Cercosporella herpotrichoides gegenuber Benomyl. Zeitschrift fur Pflanzenkrankheiten und Pflanzenschutz. Accessed Oct. 9, 2018.
- Robbertse, B., Campbell, G.F., and Crous, R.W. 1995. Revision of Pseudocercosporella-like species causing eyespot disease of wheat. South African Journal of Botany. 61:43–48.
- Scott, P.R., and Hollins, T.W. 1974. Effects of eyespot on the yield of winter wheat. Annals of Applied Biology. 78:269–279.
- Sexton, Z.F., and Murray, T.D. 2016. Evaluation of fungicides to control eyespot in winter wheat, 2015. Accessed Oct. 15, 2018.
- Wetzel, H.C. III and Murray, T.D. 2010. Evaluation of fungicides to control eyespot in winter wheat in Washington, 2009. Plant Disease Management Reports 4:CF041.
- Wetzel, H.C. III and Murray, T.D. 2011. Evaluation of fungicides to control eyespot in winter wheat in Washington, 2010. Plant Disease Management Reports. 5: CF015.
- Wetzel, H.C. III and Murray, T.D. 2013. Evaluation of fungicides to control eyespot in winter wheat in Washington, 2013. Plant Disease Management Reports. 8:1.